Table of Contents
Introduction
According to the US Centers for Disease Control and Prevention (CDC), there’s “a lot of evidence” that getting a flu shot is safe for pregnant women and their developing babies. The agency strongly encourages the practice by saying that it’s “very important for pregnant women to get the flu shot.”[1]
Yet, as we saw in part one of this series, the CDC began recommending the influenza vaccine to pregnant women during any trimester despite no randomized, placebo-controlled clinical trials having been done to determine whether this is safe. To support its recommendation, the CDC instead relies on retrospective observational studies.
We also saw in part three how the CDC’s own researchers have stressed that the type of study that would be required to determine with reasonable certainty whether or not there is a causal association between influenza vaccination and serious adverse events is the randomized placebo-controlled trial. Yet because public health officials have already made up their mind about what they think is best for us and vaccination has become routine and standardized, they regard it as “unethical” to conduct such trials now! The rationale for this is that it wouldn’t be right to deny subjects the benefits of the vaccine, which is the logical fallacy of begging the question (presuming the proposition to be proven in the premise, or the petitio principii fallacy).
While no study is methodologically perfect, the randomized controlled trial is the gold standard for safety studies precisely because other study designs aren’t as well able to control for all the innumerable variables that might enter into the equation.
Retrospective observational studies have their use for testing hypotheses in situations where scientists just aren’t able to control for variables, but such studies can only help determine whether there might be an association between a medical intervention and hypothesized adverse outcomes; they can’t prove or disprove causation. There are too many variables that would need to be controlled in order to do that to any reasonable degree of certainty.
Furthermore, while such studies can offer guidance for the direction of further research, they aren’t very useful for detecting rare unforeseen harms. They don’t well enable researchers to detect harms that they aren’t looking for.
An observational study finding an association between vaccination and an adverse outcome does not prove that the factor caused the outcome. Likewise, a finding of no association does not prove that a causal relationship does not exist. Due to the inability to control variables, observational studies are prone to selection biases that can render their findings invalid.
We’ve already seen an example of this in part two, with the selection bias inherent in the studies that the CDC has relied upon to support its claim that the influenza vaccine greatly reduces mortality among the elderly. The “healthy user” bias in these studies is an example of a failure to control for a certain variable—in this case, the difference in vaccination rates between elderly people who are in relatively good health compared to those who are frail and more likely to die soon. (It wasn’t that the vaccinated were less likely to die during a given flu season, but that those who are more likely to die are less likely to get a flu shot.)
As we saw in part one, when Aaron E. Carroll in the New York Times advocated the CDC’s flu shot recommendations, he was telling pregnant women, too, to get vaccinated. He was, in other words, parroting the CDC’s implicit message that we can trust that observational studies are methodologically robust enough to rule out the possibility, with a high degree of confidence, that vaccination could cause harm to the expectant mother or her child.
Yet just a few months earlier, Carroll had reassured the public that observational studies finding a link between alcohol consumption and cancer aren’t determinative and suggested that more randomized controlled trials are needed to establish what harms and benefits are associated with drinking! As he advised Times readers in that case, “Don’t give too much weight to observational data.”[2]
So why should we treat the findings of observational studies with healthy skepticism when it comes to the risks of drinking alcohol, but accept their findings as determinative when it comes to the risks of vaccinating pregnant women? Why should we forego our skepticism when it comes to the lives and health of entire future generations of children?
Just as this type of study can’t establish causation for an observed association, neither can they establish a lack of a causal association between vaccination and possible long-term harms. They aren’t designed to.
Carroll’s credulous acceptance of the observational studies that the CDC relies upon to support its claims is another good example of the kind of institutionalized cognitive dissonance that exists when it comes to the practice of vaccination. It has become a religion, and we are supposed to believe in the safety and effectiveness of vaccines as a matter of faith.
As another example of the logical inconsistency, in the same Times article reassuring readers that they can continue enjoying alcohol in moderation without worrying that it’s going to cause cancer, Carroll offered us the additional lesson: “Don’t cherry-pick. That is, don’t focus only on some studies, or only some results. Review all the evidence for the most holistic picture possible. . . . Acknowledge the harms, as well as the benefits, of recommendations.”[3]
Yet when it comes to vaccines, we are told by the government and media that the science is settled and that there’s nothing for us to question. We are dogmatically told that “vaccines are safe and effective”, and anyone who dares to criticize or dissent from public vaccine policy is derogatorily labeled an “anti-vaxxer”, their legitimate concerns mockingly dismissed instead of substantively addressed. Within the narrow confines of the mainstream discourse, to dare to question the wisdom of government policies with respect to vaccination is practically equivalent to committing heresy.
When it comes to vaccines, we are not supposed to concern ourselves with the methodological weaknesses of the kinds of studies the CDC relies on to support its flu shot recommendation for pregnant women. We are not supposed to notice that the CDC’s statement that “there’s a lot of evidence” that it’s safe to vaccinate pregnant women also implies that there’s at least some evidence that it is not.
And this careful wording of the CDC’s statement is understandable, given the fact that the agency’s own researchers have found an association between vaccination of pregnant women and a significantly increased risk of miscarriage.
This is the fourth installment of a multi-part exposé demonstrating how the government and media routinely and systematically deceive the public about the safety and effectiveness of the influenza vaccine. In this installment, you will learn:
- How the CDC’s own researchers have found that repeated annual vaccination of pregnant women may increase their risk of miscarriage;
- How if pharmaceutical companies made the same claims that the CDC makes about the safety of vaccinating pregnant women, they could be sued for fraud;
- How the CDC grossly mischaracterizes the findings of studies it cites to support its recommendation that pregnant women get a flu shot;
- How the CDC places the goal of maintaining or increasing vaccination rates above the goal of bettering public health;
- How the CDC deliberately misinforms the public about the toxicity of thimerosal, the mercury-based preservative that’s used in some flu shots;
- How, until thimerosal’s removal from most vaccines in the early 2000s, the CDC’s routine childhood vaccine schedule was exposing children to cumulative levels of mercury that exceeded the government’s own safety guidelines;
- How the CDC maintains that the type of mercury used in vaccines passes through the body harmlessly despite its own cited sources acknowledging that it is a known neurotoxin that can be transported across both the placental and blood-brain barriers and accumulate in the brain;
- How the CDC claims that science has proven that vaccines do not cause autism despite the biological plausibility of the hypothesis that vaccines can cause autism in subpopulations of genetically susceptible individuals and the absence of studies properly designed to actually test that hypothesis;
- How the only study designed to investigate whether getting a flu shot during pregnancy might increase the risk of having a child with autism did find an association, and why the statistical method the study authors used to make that association disappear was invalid.
- How studies have established that activation of the maternal immune system during pregnancy and the resulting inflammatory response is a risk factor for the development of autism in offspring.
- How, when it comes to vaccines, the mainstream corporate media refuse to serve the public by doing journalism and instead dutifully serve the state by colluding with the government to keep the public misinformed about what the science actually says about vaccine safety and effectiveness.
If you have not already, please be sure to read the first three installments of this series because they contain important contextual information that you will need in order to get the most benefit out of reading this fourth installment:
Part 1: Should You Get the Flu Shot Every Year? Don’t Ask the New York Times.
Part 2: How the CDC Uses Fear and Deception to Sell More Flu Vaccines.
Part 3: How You’re Being Lied to about the Risks of Getting a Flu Vaccine Annually
I am presenting the information in this third part of the series on the assumption that you already have the knowledge provided in the first three essays. So with that understood, let’s proceed.
How Vaccination of Pregnant Women May Increase the Risk of Miscarriage
We learned in part three of this series how the government has granted broad legal immunity to pharmaceutical corporations against vaccine injury lawsuits, as well as how the 1986 law that granted this immunity also established the Vaccine Adverse Events Reporting System (VAERS). As the government itself has acknowledged, one of the problems with VAERS is that only a tiny fraction of adverse events following vaccination are actually reported. Nevertheless, researchers analyzing VAERS data have identified areas of serious concern.
Among such concerns is the possibility that getting a flu shot during pregnancy may increase the risk of having a miscarriage.
In 2013, a study was published in Human & Experimental Toxicology by a former CDC researcher named Gary S. Goldman, who looked at VAERS data and found that women who received both the seasonal and the pandemic H1N1 flu shot during the 2008 – 2009 flu season had a significantly higher risk of having a miscarriage. Expressing concern that some formulations of the influenza vaccine expose the fetus to toxic mercury, Goldman hypothesized that “a synergistic fetal toxicity likely resulted from the administration of both the pandemic (A-H1N1) and seasonal influenza vaccines during the 2009/2010 season.”[4]
Another study looking at this question was published in 2017 in the journal Vaccine. With CDC funding and the participation of CDC researchers, the authors set out to examine whether it was safe for pregnant women to get a flu shot specifically during their first trimester. While the CDC publicly assures that it is safe for pregnant women to get vaccinated during any trimester, the researchers pointed out in their study that “evidence of safety in early pregnancy is limited”. Prompted by the need to address the findings of Goldman’s study, the CDC researchers ended up confirming his finding of a statistically significant association between vaccination during pregnancy and fetal loss.
Specifically, what the CDC’s own data showed was that women who received an inactivated influenza vaccine during the study period, from 2010 to 2012, had twice the risk of spontaneous abortion within twenty-eight days of receipt than women who did not receive a flu shot. Looking just at the 2010 – 2011 season, vaccinated women had a 3.7 times greater risk of having a spontaneous abortion. However, neither of these findings were statistically significant, meaning that the perceived higher risk could very well be just due to chance.
But that’s not all that their data showed. In 2009, the Influenza A(H1N1) pandemic vaccine was introduced, seasonal influenza vaccines had since included a pandemic H1N1 antigen component (pH1N1). The researchers additionally found that vaccinated women who had also received a pH1N1-containing influenza vaccine in the prior season had a statistically significant 7.7 times higher risk of spontaneous abortion.[5]
To summarize, the CDC researchers found that pregnant women vaccinated with a pH1N1-containing flu shot who had also been vaccinated with a pH1N1-containing flu shot in the prior year were nearly eight times as likely to have a miscarriage, and this finding cannot be dismissed as merely coincidental.
As the authors of this observational study noted, their data do not prove that the cause of the higher rate of miscarriage among vaccinated women was the influenza vaccine. There might be some other variable that could explain this, such as some unhealthy behavior more common among women who choose to get vaccinated during pregnancy than those who avoid doing so. Nevertheless, it is certainly an alarming finding for which the study authors offered no other obvious explanation. Certainly, the public should be made aware of this science so as to be able to make in informed choice.
Yet on the CDC’s webpage “Pregnant Women & Influenza (Flu)”, the same page where the agency says there’s “a lot of evidence” that its safe for pregnant women to get a flu shot and urges them strongly to do so, there is no mention that some versions of the vaccine contain the mercury-based preservative thimerosal and no mention of the concerns about a found association between vaccination and an increased risk of miscarriage.[6]
The CDC’s Criminal Recklessness
The US government has recommended that pregnant women receive an influenza vaccine for more than half a century. In October 1960, the US Public Health Service issued a statement by the Surgeon General that included pregnant women among population groups that “should be routinely immunized each year”.[7]
In a push to increase vaccination rates, this longstanding recommendation has in more recent decades been formalized by the CDC. In 2000, the agency’s Advisory Committee on Immunization Practices (ACIP) officially recommended vaccination of pregnant women during their second or third trimester. In 2004, the ACIP expanded its recommendation to include vaccination of pregnant women during any trimester.[8]
As we will examine in close detail, the CDC conveys the message to pregnant women that the safety of getting a flu shot annually has been scientifically demonstrated. Yet under the classification system used by the US Food and Drug Administration (FDA) for mandated labeling of all drugs to describe their safety and effectiveness during pregnancy, all licensed inactivated influenza vaccines on the market are categorized either as products for which “there are no adequate and well-controlled studies in pregnant women” or “there are no adequate and well-controlled studies in humans”![9]
As a 2012 review published in the American Journal of Obstetrics & Gynecology noted, “prelicensure data on influenza vaccine safety and effectiveness during pregnancy is virtually nonexistent because of strict research guidelines that govern the participation of pregnant women”. (Emphasis added.)
This raises an obvious question: If it is considered unethical to include pregnant women in clinical vaccine safety trials, how is it not also unethical to recommend that all pregnant women be vaccinated in the absence of clinical trials demonstrating that this is safe? In what way does the CDC’s flu shot recommendation not treat the population of pregnant women in the US as the subjects of a mass uncontrolled experiment without their informed consent?
The review further explained that “data from observational studies do not reach the standard for studies that are considered in the determination of the Food and Drug Administration pregnancy categories.” Hence, “the current classification of influenza vaccines is indicative of a lack of available data to demonstrate vaccine safety in pregnancy”.[10] (All emphasis added.)
For the CDC to publicly broadcast the message to pregnant women that science has demonstrated that it’s safe for them to get a flu shot even during the first trimester of pregnancy is not just dishonest and reckless. It is criminally irresponsible.
Consider that if a pharmaceutical company lied in this way to consumers, falsely claiming that science had demonstrated their product’s safety despite clinical safety trials never having been done, and their product was found to cause such a serious harm as fetal loss, assuming that justice is done, criminal fraud charges would be brought and responsible corporate executives would go to prison.[11]
Recall from part one that avoiding such criminal charges is precisely why all influenza vaccine manufacturers issue clear warnings against vaccinating pregnant women right on their own products’ package inserts.
For example, GlaxoSmithKline’s package insert for its Fluarix vaccine states that “Safety and effectiveness of FLUARIX have not been established in pregnant women or nursing mothers.”[12] (Emphasis added.)
To reiterate, if a pharmaceutical corporation made the same claims in their package inserts that the CDC makes in its public relations messaging, they would be violating federal laws and regulations requiring adequate warnings about the risks of using their products.
It would also be one of only two situations in which a vaccine manufacturer could be sued in civil court for vaccine injury, given the government’s granting of legal immunity under the National Childhood Vaccine Injury Act of 1986 and the Supreme Court’s upholding of near total immunity in its 2011 decision that it’s the “unavoidability” of vaccine injuries that establish “a complete defense” against injury lawsuits, provided that the vaccine is prepared according to manufacturing specifications and accompanied with adequate warnings. (For more on this legal immunity, see part three.)
It’s yet another illustration of how the CDC’s fundamental goal is not to attain better public health outcomes, but to sustain its existing policy of attaining high rates of vaccination. And it does not logically follow that the best way to achieve the former is by doing the latter. The benefits must be demonstrated to outweigh the harms, and that requires doing science that neither the pharmaceutical companies nor the government are willing to do.
The criminality of the CDC’s public messaging becomes all the more obvious in light of how it also recklessly fails to advise pregnant women to get thimerosal-free versions of the flu shot, in order to avoid unnecessarily exposing their vulnerable fetus to neurotoxic mercury.
How the CDC Mischaracterizes Its Own Cited Studies
“Protect yourself and your baby by getting your flu shot!”
That’s the message conveyed at the time of this writing by a large banner on the CDC’s webpage “Pregnant Women & Influenza (Flu)”. Here, the CDC tells pregnant women that they “should get a flu shot”; that it is “very important for pregnant women to get the flu shot”; that women should do so “during any trimester of their pregnancy”; and that “There is a lot of evidence that flu vaccines can be given safely during pregnancy; though these data are limited for the first trimester.”
As previously noted, nowhere on the page does the CDC mention the fact that some versions of the flu shot contain thimerosal. Hence, also, nowhere on the page does the CDC advise pregnant women to get thimerosal-free vaccines.[13]
The CDC does, however, provide a link for more information. That link goes to another page on the CDC’s website, titled “Flu Vaccine Safety and Pregnancy”. This page acknowledges the CDC’s own finding that “women in early pregnancy who received two consecutive annual flu shots during 2010-11 and 2011-12, did have an increased risk of miscarriage in the 28 days after receiving the second vaccine. There is an ongoing investigation to study this issue further. CDC will analyze and consider additional findings as soon as they become available.”[14]
There is a link to another page, “Flu Vaccination & Possible Safety Signal”, which provides more information and emphasizes that the study “does not prove that flu vaccine was the cause of the miscarriage.” At the time of this writing, it also states that this is still being investigated, with results “anticipated in late 2018 or 2019.”[15]
The page still says this even though the study has been completed, and its findings were presented to the ACIP in February 2019. Reportedly, it was an observational study using VSD data of women aged 18 to 44 that considered the 2012–2013, 2013–2014, and 2014–2015 flu seasons. The findings were presented to the ACIP by one of its authors, James Donahue, who stated that their findings “support current recommendations for vaccination for pregnant women in any stage of pregnancy.”[16]
Another of the study’s authors was Dr. Edward Belongia, who told CNN, “This is a very definitive study for a recent, relevant time period of flu and should remove all doubts a woman might have about whether it is safe to be vaccinated during pregnancy.”[17]
Evidently, however, the study has not yet been published in any peer-reviewed journal. As noted, the CDC webpages continue to state that the study is forthcoming, and a search on the government’s database of published studies, PubMed.gov, shows that the last published study including Donahue and Belongia as coauthors was the 2017 CDC-funded study finding an association between consecutive annual administration of pH1N1-containing vaccines and a sevenfold increased risk of miscarriage.[18]
Furthermore, there is a very good reason to doubt the CDC’s characterizations of studies it cites to support its claims of vaccine safety, which is that the CDC systematically misrepresents the science to support its policy.
We have already seen numerous examples in earlier installments of this series. The “Flu Vaccine Safety and Pregnancy” page of the CDC’s website provides several more. “There is a large body of scientific studies”, the CDC states, “that supports the safety of flu vaccine in pregnant women and their babies.” To support this statement, the CDC cites seven observational studies, three of which looked at data from VAERS and the rest at data from the Vaccine Safety Datalink (VSD).[19] The “Flu Vaccination & Possible Safety Signal” page cites five of the same studies.[20] (For discussion about some of the major problems with VAERS and the VSD, see part three).
The problem with this is that none of these studies support the CDC’s claim that science has shown that getting a flu shot during pregnancy is safe for both the expectant mother and her unborn child. To illustrate the low quality of the CDC’s supposed evidence, as well as how the agency mischaracterizes its own cited sources, let’s briefly examine each.
Moro et al., April 2011
The first VAERS study that the CDC cites as proof of the safety of vaccinating pregnant women, authored by a team of CDC researchers and published in the American Journal of Obstetrics and Gynecology in February 2011, found that the most commonly reported adverse event following administration of the trivalent inactivated influenza vaccine to pregnant women was spontaneous abortion. But even for the years 2008 – 2009, during which the highest reporting rate of spontaneous abortions occurred, the rate of miscarriage among vaccinated women “was lower than published rates of miscarriage in the general pregnant population.” (Emphasis added.)
Yet the CDC researchers did not claim that this finding shows that the vaccine is associated with a decreased risk of miscarriage. On the contrary, they acknowledged that this was “likely due to underreporting of this event in VAERS”. They explicitly concluded that “we cannot make any inferences regarding the rate of this event following vaccination.” Once again in their own words, their estimated rates of spontaneous abortion “may be underestimating the true rates of miscarriage in pregnant women who received influenza vaccine.”[21] (Emphasis added.)
In other words, we cannot infer from the results of this study that vaccinating pregnant women is safe and does not increase their risk of having a miscarriage.
So, as you can see, while the CDC claims that this study shows that vaccinating pregnant women is safe, in fact it does not.
Moro et al., November 2011
The second VAERS study cited by the CDC was written by largely the same team of CDC researchers and published in the same journal in November 2011. It looked specifically at adverse events reported after administration of the 2009 pandemic H1N1 monovalent inactivated influenza vaccine.
The authors noted that few studies had considered the safety of vaccinating pregnant women specifically during the first trimester—which their own study was also not designed to do—and stressed that this was “a gap that should be addressed in future vaccine safety research.”
They also noted that no studies had been conducted to determine the safety of vaccinating pregnant women with the pandemic H1N1 vaccine before the CDC recommended it for them. Instead, the “assumption” was made by the CDC that its safety would be “similar to that of seasonal influenza vaccine”.
Continuing, what their data counterintuitively showed was that rates of spontaneous abortions and stillbirths reported after H1N1 vaccination were “several orders of magnitude lower than the expected rates of fetal losses in the general population of pregnant women”. (Emphasis added.)
Once again, they did not conclude that the vaccine is associated with a reduced risk of miscarriage. Instead, the CDC researchers acknowledged that this result was simply indicative of the problem of underreporting to VAERS. In their own words, “There is underreporting to VAERS in general, and the proportion of AEs [adverse events] following immunization among pregnant women that are reported to VAERS is unknown.”[22]
So, once again, as you can see, the CDC is citing a study that it claims demonstrates the safety of vaccinating pregnant women but which in fact does not.
Moro et al., December 2017
The third VAERS study cited by the CDC was published in Drug Safety in December 2017. It was conducted by five researchers from the CDC and one from the FDA who expressed concern that pregnant women were reluctant to get the flu shot due to worries about its safety. An implicit aim of their study was to address this “barrier to vaccine uptake” by using VAERS data “to assess the safety of seasonal influenza vaccine in pregnant women and their infants whose reports were submitted to VAERS during 2010–2016.”
In other words, the government researchers set out to show that the vaccine is safe and so designed a study that they could cite to reassure the public. Once again, the means to that end was to examine VAERS data. Once again, they found that spontaneous abortion was the most frequently reported pregnancy-related adverse event, comprising 11.4 percent of those reported. They noted that miscarriages are known occur in up to 22 percent of pregnancies among women aged thirty-four or older. During the 2009 H1N1 pandemic, however, miscarriages accounted for 41 percent of reported adverse events for pregnant women who’d received the pandemic influenza vaccine. The proportion of stillbirths reported was also increased.
The authors acknowledged that, prior to the 2009 pandemic, pregnancy reports after influenza vaccination “had been sparsely reported to VAERS”, but then spiked during the pandemic season. But they dismissed the spike as being attributable to “over-reporting bias surrounding publicity around certain events.”[23]
But that explanation doesn’t actually make sense. To support it, they cite a study discussing the possibility that if the FDA issues a safety alert about a drug, it can result in an increase in reporting about a given adverse event. But that study found that FDA alerts typically did not result in what’s described as “stimulated” reporting.[24] Furthermore, the CDC researchers failed to identify any FDA alert or media reporting of a potential safety issue that could possibly explain stimulated reports of miscarriages and stillbirths due to influenza vaccination!
Additionally, their assumption that the spike was due to “over-reporting” simply ignored the CDC’s own previous findings of significant underreporting of serious adverse events. Obviously, if there was no increased awareness of a possible safety issue, their findings strongly suggest a very serious problem with one or more of the vaccines used that season. And even if reporting had been stimulated in 2009, the possibility would remain that the spike more closely reflected the true rate of adverse events following vaccination.
It was by such means that the authors dismissed the possibility that the increased proportion of miscarriage reports might indicate a true safety signal related to the use of either or both the seasonal and the pandemic flu vaccines.
Incongruently, with respect to reports of major birth defects, they did acknowledge a “substantial level of underreporting”. They also acknowledged that their study’s limitations included the fact that VAERS “may be prone to biased reporting (over or underreporting)”. Their main conclusion was not that their findings demonstrate the safety of vaccinating pregnant women, but simply that “no new or unexpected patterns in maternal or fetal outcomes were observed”, which depended on their assumption that the spike in reports and increased proportion of miscarriages and stillbirths reflected over-reporting and not a true safety signal.
It should not surprise us, of course, that government researchers who set out to find no association between vaccination and adverse events end up concluding that there’s no evidence of harm, even when it means simply choosing not to see what their own data are showing them. A finding of no harm is also not at all surprising given the known problem of underreporting and the fact that, as the study’s authors explicitly conceded, “it is not possible to calculate the incidence or prevalence of” adverse events using VAERS data.[25]
In other words, their findings do not establish one way or the other whether it is safe to vaccinate pregnant women. This study, like the other two VAERS studies, is practically worthless and does not support the CDC’s claim. On the contrary, the study’s actual findings, as opposed to the authors’ illogical conclusion, strongly suggest a serious problem with one or both of that season’s influenza vaccines.
Irving et al., January 2013
The first VSD study cited by the CDC was conducted by researchers from the CDC and the health care company Kaiser Permanente. It was funded under a contract from the CDC. Half of its twelve authors reported having previously received research funding from pharmaceutical companies. This included James Donahue and Edward Belongia, both of whom reported previously receiving research funding from Medimmune, LLC, the manufacturer of FluMist, a live-virus, intranasally administered influenza vaccine.
Published in Obstetrics & Gynecology in January 2013, it was an observational study comparing rates of exposure to influenza vaccines between a group of women who’d had a spontaneous abortion during the autumn of 2005 or 2006 and a matched group who’d had a live birth. Describing the evidence supporting the safety of influenza vaccination in early pregnancy as “sparse”, their own finding was that those who had a spontaneous abortion had a slightly higher odds of having received an influenza vaccine within 28 days prior, but the increased risk was “not statistically significantly”.
As they explicitly conceded, “our sample size was not large enough to rule out any increase in risk.” They hence called for “further research on the safety of influenza vaccine in pregnant women”.[26]
The CDC states that this study “found no increased risk of miscarriage among pregnant women who received flu vaccines in the 2005-06 or 2006-07 flu seasons.”[27] An honest summation would be that it did find an increased risk of miscarriage but did not include enough subjects to determine whether this finding was more likely due to chance or a real association, and so further studies are urgently required.
This study, too, simply does not support the CDC’s claim that vaccinating pregnant women has been proven safe, which further demonstrates how the CDC has no interest in honestly informing the public what the science says.
Kharbanda et al., September 2013
Of the second VSD study cited, the CDC says it “found no increased risk for adverse obstetric events (like chorioamnionitis, pre-eclampsia, or gestational hypertension) for pregnant women who received the flu vaccine from 2002 to 2009 when compared to pregnant woman who were not vaccinated.”[28]
This study was published in Obstetrics & Gynecology in September 2013. Once again, the impetus for the study was that “Fears regarding the safety of influenza vaccine during pregnancy remain a persistent barrier to vaccine uptake.” Although the study population included women who’d had a miscarriage, its authors did not compare birth outcomes. Instead, they “focused on pregnancy-related comorbidities that directly affect maternal health”. Hence this study does not support the CDC’s claim that it is safe for the developing fetus for pregnant women to get a flu shot.
Furthermore, the study actually undermines the CDC’s use of VAERS studies to support its claims, since the authors highlighted such studies’ shortcomings, including the facts that VAERS is “prone to underreporting” and does not provide a control group from which to make comparisons.
Contrary to the CDC’s characterization, the study did find an increased risk for one maternal outcome: a statistically nonsignificant higher risk for vaccinated women of chorioamnionitis (inflammation due to bacterial infection of the membranes surrounding the fetus and amniotic fluid). The authors acknowledged that a “potential mechanism” for such an increased risk “would be the measurable vaccine-induced inflammatory response”, but went on to express reasonable “doubt” that their results “represent a true association.”
Finally, the authors acknowledged that their findings were subject to “residual unmeasured confounders and healthy vaccinee bias”, as had been “observed frequently in nonpregnant populations.” (Emphasis added.)
Indicating the possibility of this selection bias in their own study, they noted that, for example, “those vaccinated during pregnancy had more medical encounters both before and after vaccination, possibly reflecting increased health-seeking behaviors.” Another weakness of the study potentially biasing their results toward a finding of no association was the possibility that some women included in the study as “unvaccinated” had received the vaccine at a pharmacy or their workplace instead of from their health care provider.[29]
In short, what this study’s findings actually support is not the safety of vaccinating pregnant women, but the urgent need to control for the known problem of healthy vaccinee bias by conducting randomized placebo-controlled trials to determine the safety for both the pregnant vaccinee and her unborn child.
Nordin et al., May 2014
The third VSD study the CDC cites was published in The Journal of Pediatrics in May 2014. It compared the rates of premature delivery or being small for gestational age between women who received the flu shot and women who didn’t during the 2004–2005 and 2008–2009 flu seasons. No increased risk was found between either of these two outcomes and vaccination.
No previous study, its authors noted, had examined these two outcomes by looking at a large cohort of women vaccinated during their first trimester. They acknowledged the known problem of healthy vaccinee bias and tried to account for it by matching women for comparison who, “based on available data, had an equal likelihood of being vaccinated”. But they acknowledged that their ability to control for such bias was limited and that “it is likely that other unmeasured confounders differed between the vaccinated and unvaccinated women.”
“Finally,” in the words of the authors, “this sample was limited to live births, and focused on 2 specific perinatal outcomes. Other important birth outcomes were not analyzed. Inclusion of stillbirths or evaluating risks for spontaneous abortion would require alternative analytical approaches and additional data not available for this large cohort at the time of this study.”[30]
Thus, this study does not support the CDC’s assurances that pregnant women are not putting themselves at an increased risk of having a stillbirth or miscarriage if they get a flu shot. Nor does it support the CDC’s claim that this practice does not increase the risk of neurodevelopmental harm to the vulnerable fetus. Once again, it simply illustrates the necessity of doing randomized controlled trials in order to better control for the recognized problem of healthy user bias.
Kharbanda et al., August 2017
The final study cited by the CDC also used VSD data. Published in The Journal of Pediatrics in 2017, it was also funded by the CDC. Four of its thirteen authors disclosed having previously received research funding from one or more pharmaceutical companies. Their study population was comprised of women who’d had live births between 2004 and 2013, the purpose being to compare rates of major birth defects between women who’d received a flu shot during their first trimester and women who’d either had a flu shot later during their pregnancy or not at all.
A secondary analysis compared the former cohort with women who’d received no flu shot during any trimester, which resulted in similar findings of no increased risk of birth defects associated with vaccination during the first trimester.
Among the limitations acknowledged by the authors were that infants in their study “were likely at lower risk than the general US population for having a major structural birth defect” and that their study was not designed to compare rates of stillbirths or spontaneous abortions.[31]
Summation of CDC’s Cited Studies
The CDC claims that each of these studies show that it is safe to vaccinate pregnant women, but they do not. They cannot demonstrate the safety of this practice because they were not properly designed to.
The VAERS studies suffer from the problem of underreporting of adverse events and lack of a comparison group and so are practically worthless.
The VSD studies examined only limited outcomes and are prone to selection bias. Despite it being the most frequently reported adverse event after influenza vaccination, only one of these studies examined the question of whether the flu shot might increase the risk of miscarriage, and it did find an increased risk that may only have been statistically nonsignificant because it was admittedly underpowered.
Furthermore, not one of these studies was designed to determine whether vaccination during pregnancy might cause neurodevelopmental harm to the fetus, such as an increased risk of autism.
If the CDC’s primary interest was in bettering public health, it would not be citing these studies to reassure the public about the safety of vaccinating pregnant women; it would instead be sounding the alarm that the postmarketing surveillance system in place to detect harms from vaccines is totally inadequate and that randomized placebo-controlled trials are urgently required to determine whether it is safe for pregnant women to be vaccinated.
Mercury Toxicity and the CDC’s Deliberate Failure to Inform
As the CDC knows perfectly well, the kind of study that would need to be done to properly assess the risks versus benefits of vaccination is the randomized placebo-controlled trial, which government agencies refuse to do on the grounds that it would be “unethical” to deprive the control group of the presumed benefits of the vaccine.
Of course, the CDC has an inherent conflict of interest in conducting any safety studies at all. After all, since the CDC itself bears significant responsibility for any harms caused by vaccinating according to its recommendations, it has a strong incentive to conduct studies that are not well designed to detect serious harms from vaccines. Indeed, poorly designed studies are exactly what we should expect from the CDC. And that’s just what we see.
To further illustrate, among the aforementioned studies cited by the CDC to support its claim that the flu shot is safe for pregnant women, none considered the question of whether vaccination with thimerosal-containing flu shots is safe for the developing fetus. Indeed, not one of these studies distinguished between thimerosal-free flu shots and those that do contain this mercury-based preservative.
On the same “Flu Vaccine Safety and Pregnancy” webpage, as already noted, the CDC does disclose the finding in the 2017 Vaccine study of an increased risk of miscarriage within 28 days of vaccination. But it stresses that this observational study does not show that vaccination caused miscarriages and that it continues to recommend vaccination of pregnant women during any trimester “because flu poses a danger”.
This, of course, is a non sequitur fallacy: it does not logically follow from the fact that there are risks associated with getting the flu that therefore it is wise for pregnant women to get vaccinated, and particularly not with a mercury-containing vaccine.
Near the bottom of this page, the CDC does disclose that some versions of the flu shot contain thimerosal. It does not explain, however, that the concern about thimerosal is the fact that it exposes both the expectant mother and the vulnerable fetus to the known neurotoxicity of ethylmercury.
The word “mercury”, in fact, appears nowhere on the webpage.
Here’s all that the CDC says about it: “Studies have shown the small amount of thimerosal in vaccines does not cause harm. There is thimerosal-free flu vaccine available for people who want to avoid thimerosal.”
Apart from not explaining what thimerosal is, the CDC does not actually advise pregnant women to avoid it. Beyond that, all the CDC offers is another link to another page on their website.[32]
That linked page, titled “Thimerosal in Flu Vaccine”, does inform that thimerosal “is an ethyl mercury-based preservative used in vials that contain more than one dose of a vaccine (multi-dose vials) to prevent germs, bacteria and/or fungi from contaminating the vaccine.”[33] (The use of a preservative in multi-dose vaccine vials is actually required by the FDA.[34])
However, the CDC once again does not inform that ethylmercury is a known neurotoxin. Instead, it simply tells the public that it “has a record of being very safe”, and that “many studies” have shown “no evidence of harm caused by the low doses of thimerosal in vaccines.”
The CDC provides no sources, apart from links to two more CDC webpages, respectively titled “Thimerosal in Vaccines” and “Frequently Asked Questions about Thimerosal”.[35]
The “Thimerosal in Vaccines” page states, “There is no evidence of harm caused by the low doses of thimerosal in vaccines, except for the minor reactions like redness and swelling at the injection site.”[36]
But that statement is practically meaningless, given the fact that, as a 2001 review by FDA researchers admitted, “no controlled studies have been conducted to examine low-dose thimerosal toxicity in humans”. (Emphasis added. It remains true, to my knowledge, that no prospective controlled studies in humans have been conducted to determine the risk.)
Published in the journal Pediatrics, the FDA review conceded that “we cannot exclude the possibility of subtle neurodevelopmental abnormalities from the cumulative exposure to thimerosal in vaccines”.[37]
So why does the CDC publicly convey the message that we can exclude that possibility?
Whatever the explanation, what is absolutely incontrovertible is that there is a huge and irreconcilable disparity between what the CDC says science says about the safety of vaccinating pregnant women and what the science actually tells us.
Why Thimerosal Was Really Removed from Most Childhood Vaccines
After meaninglessly claiming “no evidence of harm”, the CDC’s “Thimerosal in Vaccines” page continues: “However, in July 1999, the Public Health Service agencies, the American Academy of Pediatrics, and vaccine manufacturers agreed that thimerosal should be reduced or eliminated in vaccines as a precautionary measure.”[38]
What the CDC doesn’t say is that the reasons for this decision were that: 1) it was economically viable for the vaccine manufacturers to do so; and 2) it had become known that, as the CDC continued adding more thimerosal-containing vaccine doses to the schedule, no government agency ever bothered to calculate the cumulative levels of mercury that children were being exposed to, and when the FDA finally got around to doing so, it determined that the levels exceeded the safety guidelines determined by the US Environmental Protection Agency (EPA).[39]
The CDC also does not explain that the EPA’s safety guideline was established for ingested methylmercury, not injected ethylmercury, because there are no established safety guidelines for exposure to ethylmercury (whether ingested or injected).
But as the FDA researchers acknowledged in their Pediatrics review, “similar toxicological profiles between ethylmercury and methylmercury suggest that neurotoxicity may also occur at low doses of thimerosal”. (Emphasis added.)
The Pediatrics study also reported the FDA’s finding that the cumulative exposures exceeded the EPA’s recommendations.[40]
The reason the FDA finally got around to doing the calculations was because in 1997, Congress passed the FDA Modernization Act, a provision of which required the FDA to compile a list of mercury-containing drugs on the market. The products included in the industry’s responses to the FDA’s inquiries included more than thirty licensed vaccines.
When the FDA’s Center for Biologics Evaluation and Research (CBER) in 1999 did the calculations showing mercury exposure in excess of the EPA’s guidelines, government officials waffled over what to do about it. In an email to the acting director of the CDC’s National Vaccine Program Office, Martin Meyers, the director of the FDA’s Division of Viral Products, Dr. Peter Patriarca, opined that to call for the removal of thimerosal from vaccines would
raise questions about FDA being ‘asleep at the switch’ for decades by allowing a potentially hazardous compound to remain in many childhood vaccines, and not forcing manufacturers to exclude it from new products. It will also raise questions about various advisory bodies regarding aggressive recommendations for use. (We must keep in mind that the dose of ethylmercury was not generated by “rocket science”. Conversion of the percentage thimerosal to actual micrograms of mercury involves ninth grade algebra. What took the FDA so long to do the calculations? Why didn’t CDC and the advisory bodies do these calculations when they rapidly expanded the childhood immunization schedule?)[41] [Emphasis added.]
While acknowledging exposure in excess of the EPA’s guidelines, the FDA researchers in their Pediatrics study also claimed that the maximum cumulative exposure of 187.5 micrograms of mercury did not exceed the guidelines established by either the Agency for Toxic Substances and Disease Registry (ATSDR) or the FDA.
However, this, too, was deceptive. One expert whom the FDA consulted was Dr. Barry Rumack, whose calculations showed that children were indeed being exposed to levels of mercury far in excess of all three federal safety guidelines: the EPA’s, the ATSDR’s, and FDA’s own. Corresponding to when infants would receive thimerosal-containing doses of vaccines, dramatic spikes in exposure were calculated at zero, two, four, six, and twelve months of age. From the age of two months to approximately ten months, as well as from twelve months to approximately fourteen months, blood mercury levels calculated were in excess of the FDA’s suggested limit. From birth until approximately nineteen months of age, there was no time at which the mercury exposure from vaccines fell below the EPA’s suggested limit.[42]
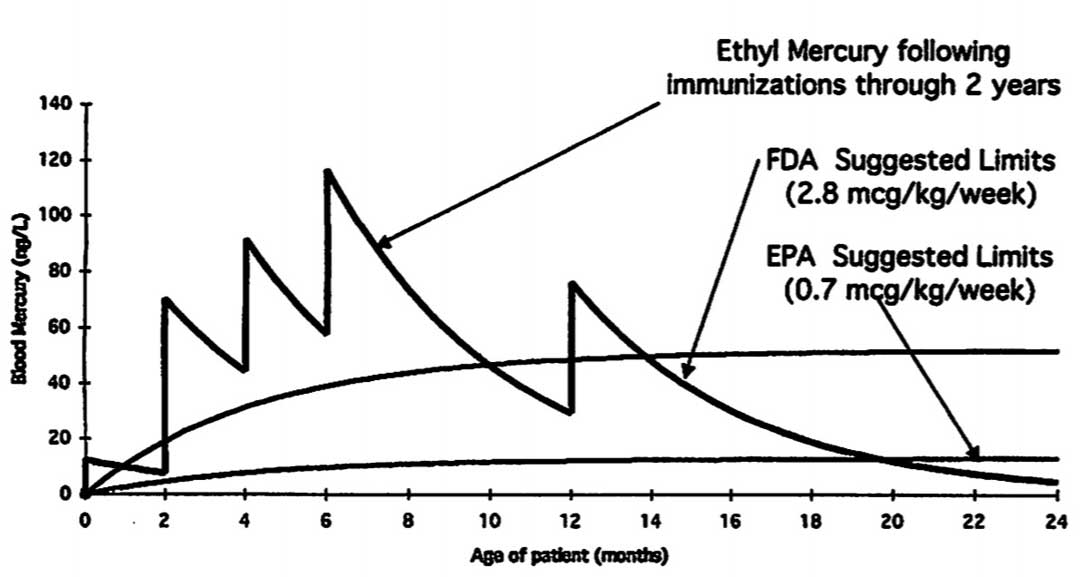
Researchers David Geier and Mark Geier also independently calculated mercury exposure from the CDC’s childhood schedule, confirming that the levels were in excess of the FDA’s own established limits. By their calculations, “the instantaneous relative excess mercury that US children received from their childhood immunizations ranged from 3.2 – 32-fold in comparison to the US FDA safety guidelines for the daily maximum oral ingestion of methylmercury.”[43]
The relatively less alarming result reported by the FDA researchers in their Pediatrics study was obtained by averaging the exposures over a six month period, which is unscientific because in reality the exposures would occur instantaneously at just a few points in time, not evenly over the duration. As Robert F. Kennedy, Jr. and Lyn Redwood have commented, “An analogy would be to compare taking two Tylenol tablets a day for a month to taking 60 Tylenol tablets in one day; the first exposure is acceptable, while the other is lethal. Using this misleading gimmick, regulators were able to report that mercury exposure levels were below FDA and ATSDR guidelines.”[44]
Another shortcoming of the FDA’s published calculation is that it failed to account for other environmental sources of mercury that infants are exposed to besides vaccines. It was therefore by design a conservative estimate of mercury exposure in the US childhood population.
In commenting on this fact in a 1999 email to the director of the FDA’s Office of Vaccines Research, Norman Baylor, the lead author of the Pediatrics study, Leslie K. Ball, also expressed concern that “toxicologists seemed reluctant to state any Hg [mercury] was ‘safe’”, which left public health officials open to the criticism that they were “arbitrarily designating a certain level as acceptable when there continues to be so much uncertainty about the science in this area.”[45] (Emphasis added.)
Returning once more to the CDC’s “Thimerosal in Vaccines” webpage, it’s not at all surprising that the CDC would decline to inform the public that the reason thimerosal was removed from most childhood vaccines is because the agency’s routine childhood schedule was exposing infants to mercury levels that exceeded the government’s own safety guidelines.
After all, to disclose this information might lead members of the public to reasonably conclude that they cannot trust government agencies like the FDA and CDC.
And that would be counterproductive to the CDC’s primary goal of increasing demand for the pharmaceutical industry’s vaccine products.
More CDC Deception: Ethylmercury vs. Methylmercury
A bit farther down the “Thimerosal in Vaccines” page, the CDC states that ethylmercury from vaccines “is cleared from the human body more quickly than methylmercury, and is therefore less likely to cause any harm.” Farther down still, the CDC states:
The human body eliminates thimerosal easily.
Thimerosal does not stay in the body a long time so it does not build up and reach harmful levels. When thimerosal enters the body, it breaks down to ethylmercury and thiosalicylate, which are readily eliminated.
Thimerosal has been shown to be safe when used in vaccines.
Thimerosal use in medical products has a record of being very safe. Data from many studies show no evidence of harm caused by the low doses of thimerosal in vaccines.
The only side-effects of thimerosal the CDC acknowledges are “minor reactions like redness and swelling at the injection site” and, rarely, allergic reaction.
The CDC cites no studies on this page to support its claim that the mercury in vaccines is “very safe”, including for children and pregnant women. It does provide a link to a PDF file titled “Understanding Thimerosal, Mercury, and Vaccine Safety”. That file does cite seven studies. None of these were randomized controlled trials. Six were observational studies, and the seventh was a 2004 report by the Institute of Medicine (IOM) that acknowledged the methodological limitations of observational studies; described thimerosal as a “known neurotoxin”; acknowledged that some of the mercury from thimerosal exposure “accumulates in the brain”; and admitted that “heavy metals, including thimerosal, can injure the nervous system.”[46]
How is it that a substance described uncontroversially in the medical literature as a known neurotoxin that accumulates in the brain and may cause neurological harm even at low doses can be described to the public by the CDC as “very safe” to inject into children and pregnant women because it is “readily eliminated” from the body? The simple answer is that, when the CDC tells the public that science has shown that the mercury from vaccines is harmless because it is rapidly eliminated from the body, it is deliberately lying.
In fact, what science actually tells us is precisely the opposite of what the CDC says, which we can observe once again simply by examining the CDC’s own cited sources.
Turning to the CDC’s “Frequently Asked Questions about Thimerosal” page, we find the CDC once again assuring us that the ethylmercury from thimerosal in vaccines is “safe”. Further, “Scientists have been studying the use of thimerosal in vaccines for many years. They haven’t found any evidence that thimerosal causes harm.” At the bottom the page, there’s a link to yet another webpage, which provides references from the scientific literature intended to support the CDC’s claims about the safety of thimerosal-containing vaccines.[47]
Yet the first reference listed is the very same Pediatrics study in which FDA researchers admitted that the government was responsible for exposing children to levels of mercury that exceeded the EPA safety guidelines, that ethylmercury is neurotoxic even at low doses, and that it is possible that mercury exposure from vaccines may cause neurodevelopmental abnormalities in children.[48]
The second reference is a study published in Environmental Health Perspectives in 2005, which incidentally has been described by FDA researchers as “the most relevant study on which to base a comparative assessment of infant mercury exposure or risk from thimerosal [ethylmercury] relative to MeHg [methylmercury].”[49]
This 2005 study, by Thomas M. Burbacher et al., compared the levels of mercury in the brains of infant monkeys after exposure from either ingested methylmercury or thimerosal-containing vaccines at doses relatively comparable to the infant exposure from the CDC’s childhood schedule. They confirmed that ethylmercury from vaccines is more rapidly eliminated from the blood than methylmercury, and also that less total ethylmercury enters the brain. However, they also found that once inside the brain, ethylmercury is much more readily converted to inorganic mercury, which is not as readily eliminated from the brain as methylmercury, which, like ethylmercury, is an organic form (a compound in which the mercury is molecularly bound to carbon).
That is to say, rather than being more readily eliminated, as claimed by the CDC, the form of mercury in vaccines is more persistent in the brain.
Far from concluding that their findings showed that the mercury in vaccines is safe, what Burbacher et al. actually concluded was that the government’s use of methylmercury toxicology as a reference for risk assessment from thimerosal exposure is scientifically invalid, and that the use of mercury in vaccines represents a significant cause for concern.
Noting that “a much higher proportion” of inorganic mercury was found in the brains of thimerosal-exposed monkeys, they further observed that inorganic mercury in the brain has been “associated with a significant increase in the number of microglia in the brain” and that “‘an active neuroinflammatory process’ has been demonstrated in brains of autistic patients, including a marked activation of microglia.”
In their concluding remarks, they also expressed dismay that the Institute of Medicine, in its 2004 report, implicitly recommended that further research not be undertaken to test the biologically plausible hypothesis that vaccines can cause autism. “This approach is difficult to understand,” they remarked, “given our current limited knowledge of the toxicokinetics and developmental neurotoxicity of thimerosal, a compound that has been (and will continue to be) injected into millions of newborns and infants.”
Far from supporting the CDC’s claim that science has shown that mercury exposure from vaccines is “very safe”, this study actually concluded that more research into the neurotoxic potential of thimerosal and its breakdown product ethylmercury was “urgently needed” to be able to draw any meaningful conclusions about “the potential developmental effects of immunization with thimerosal-containing vaccines in newborns and infants.”[50]
To cite just a few other studies demonstrating the falsity of the CDC’s claims about ethylmercury being rapidly and harmlessly eliminated from the body, a 2004 rodent study by researchers from the National Institutes of Health (NIH) published in Toxicology Letters similarly found that, while ethylmercury is readily eliminated from the blood, it is persistent in the tissue, the kidneys, and the brain.[51]
A team of Italian researchers published a study in Interdisciplinary Toxicology in 2012 finding that thimerosal is fifty times more toxic than methylmercury to human cells in terms of the dose required to significantly decrease cellular viability or induce cell death. As they concluded, “Our findings demonstrate that thimerosal at the concentration usually found in vaccines, affects significantly cellular viability.”[52] (Emphasis added.)
It has also been known since at least 1975 that thimerosal crosses both the blood-brain and placental barriers.[53]
A meta-analysis of studies on low-dose thimerosal exposure published in Neurochemical Research in 2011 observed that all such studies to date had found evidence of neurotoxicity.
Furthermore, no studies had considered the synergistic toxicity of thimerosal-containing vaccines being administered concomitantly with aluminum-containing vaccines. (For a discussion of the risks from aluminum, see part three. Synergistic toxicity refers to toxic effects that are not just additive, but multiplicative.)
Additionally, animal studies had shown that thimerosal exposure results in accumulation of inorganic mercury in the brain; and, at doses relevant to the exposure children receive from the CDC’s schedule, thimerosal’s demonstrated neurotoxicity “possesses the potential to affect human neuro-development.”
The cumulative exposure to thimerosal that pregnant women and infants get from vaccines, the review concluded, “is a likely risk factor for neurodevelopmental delays that has yet to be defined.” There may also be “subtle neurodevelopmental delays in susceptible infants” that “may not be perceived in routine medical examinations.”
The evidence that low doses of thimerosal from vaccines “can affect neural tissues and functions” is “unequivocal”.
Consequently, “use of thimerosal in vaccines should be reconsidered by public health authorities, especially in those vaccines intended for pregnant women and children.”[54] (All emphasis added.)
A study published in the Cambridge Journal of Developmental Origins of Health and Disease in 2013 noted that there had been growing concerns globally about the levels of mercury and other heavy metals that pregnant women were being exposed to, and particularly about the effects on the growth and development of their vulnerable fetus. It relayed the prevailing scientific consensus that there are “no safe limits for blood levels of heavy metals in pregnancy”.[55] (Emphasis added.)
A review published in 2017 in the International Journal of Environmental Research and Public Health likewise commented on the risk to the developing human fetus from exposures to mercury and other heavy metals during pregnancy. Noting that studies had found an association between the receipt of multiple influenza vaccinations and an increased risk of miscarriage, it concluded that “governmental changes and directives are clearly needed concerning vaccines and any neurotoxic content”.
Furthermore, the practice of deliberately exposing children to known neurotoxins at the levels prescribed by the CDC’s routine vaccine schedule “has to be considered dangerous, irresponsible, and certainly should be ended.”[56]
So now ask yourself: Why would the CDC not advise pregnant women strongly to get a thimerosal-free single-dose flu shot in order to avoid unnecessarily exposing their developing fetus to the known neurotoxicity of ethylmercury?
The answer, once again, is simple: the CDC has more interest in protecting its own reputation and achieving is policy goal of high vaccination rates than it does in achieving better public health.
How the Influenza Vaccine Might Increase the Risk of Autism
As we’ve seen, to support its claim that ethylmercury is “very safe” to inject into pregnant women and children, the CDC in its “Understanding Thimerosal, Mercury, and Vaccine Safety” document cites a handful of observational studies and a 2004 IOM review that actually described thimerosal as a “known neurotoxin” and acknowledged its potential to cause neurological harm in children.
On another page of the CDC’s website, it authoritatively declares, “Vaccines Do Not Cause Autism”. That page also cites the 2004 IOM review, quoting it as having concluded that “the evidence favors rejection of a causal relationship between thimerosal-containing vaccines and autism.”[57]
Yet the IOM review also noted that all of the studies finding no association were observational studies, and it explicitly acknowledged that the hypothesis that vaccines can cause autism among genetically susceptible subpopulations cannot be excluded by such studies! In the words of the IOM report, “This hypothesis cannot be excluded by epidemiological data from large population groups that do not show an association between a vaccine and an adverse outcome.”
The IOM further acknowledged that none of the studies it examined were actually designed to test that hypothesis. This, they conceded, could theoretically explain why these studies failed to find an association![58]
Also of note is the fact that all of the studies included in the IOM review only considered either one vaccine on the childhood schedule (the measles, mumps and rubella combination vaccine, or MMR) or one vaccine ingredient (thimerosal).[59] Recall from part three the acknowledgment by the IOM in its 2013 report that “studies designed to examine the long-term effects of the cumulative number of vaccines or other aspects of the immunization schedule have not been conducted.”[60]
The CDC on its “Vaccines Do Not Cause Autism” page also cites nine CDC-funded studies conducted between 2003 and 2012 that found no association between autism and either the MMR vaccine or thimerosal.[61] The PDF file listing these studies claims that they show that “thimerosal is not a toxin in vaccines”.[62]
As indicated in the 2013 IOM review, none of these studies were designed to examine the long-term effects of vaccines administered according to the CDC’s routine childhood vaccine schedule. And since none of them were designed to test the hypothesis that vaccines administered according to the CDC’s recommendations can contribute to the development of autism in children with a genetic or environmental predisposition, none support the CDC’s claim that this hypothesis has been scientifically falsified.
The CDC’s Cited Studies on Vaccines and Autism Relevant to the Flu Shot
While it’s beyond the scope of this series to examine each of these nine studies, four of them bear some relevance for the CDC’s flu shot recommendation for pregnant women, so let’s take a brief look at those, as doing so once again illustrates how the CDC grossly mischaracterizes the science.
The first was a study by William W. Thompson et al. published in the New England Journal of Medicine in 2007. The authors looked for associations between early thimerosal exposure and forty-two neurophyschological outcomes between ages seven and ten. As the authors note, their study “did not examine the possible association between autism and exposure to mercury from vaccines”. Of course, it isn’t surprising that a study that wasn’t designed to look for an association between vaccines and autism would not find one.
Apart from that, there were numerous other problems with this study. Their study population included only 1,047 children and was therefore underpowered to detect rare harms, such as if thimerosal-containing vaccines contributed to the development of autism in one in ten thousand children. Relatedly, they also failed to consider the possibility of a subpopulation of children who are at higher risk of neurological harm from mercury. In fact, they actually excluded children who might be at higher risk, such as if they had a low birth weight or had been diagnosed with encephalitis (brain inflammation). Their curious criteria for excluding children with these and other conditions was that they “could bias neuropsychological testing”—which can reasonably be interpreted as a euphemism meaning that to include them might increase the likelihood of finding associations between vaccination and symptoms of neurological harm.
Thompson et al. took into consideration prenatal exposure to mercury due to maternal vaccination as well as postnatal exposure from thimerosal-containing vaccines through the first seven months of life. But less than 11 percent of children included in their study population had recorded prenatal exposures, and only nine children had exposure due to maternal influenza vaccination. Hence their study is useless for assessing the safety of this practice. The authors also did separate analyses for prenatal and postnatal exposures rather than considering the total cumulative exposure.
Among their findings were both positive and negative associations with thimerosal, such as higher performance IQ among boys and lower verbal IQ among girls. As the authors stated, this “makes it difficult to draw general conclusions about possible effects of neonatal mercury exposure from vaccines . . . on intellectual abilities.” This also means, of course, that their findings cannot be used to support definitive statements like the CDC’s declaration that vaccines do not cause autism. Among the statistically significant associations they found was an increased risk of tics, as had also been found by previous studies.
Thompson et al. also acknowledged the finding by Burbacher et al. of “persistently low levels of inorganic mercury in the brains of monkeys exposed to ethyl mercury”, adding that “the implication of these findings for humans is not known.” This also means, of course, that their own study did not answer the question of what harms mercury accumulation might be causing the US childhood population.
Among other limitations of the study, the authors also acknowledged that their findings “may have been influenced by selection bias”. They were unable to control for “interventions, such as speech therapy, that may have ameliorated the potential negative effects of thimerosal exposure and could have biased the results toward the null hypothesis”—meaning toward a finding of no association. They also did not consider thimerosal exposure from vaccines after seven months of age.
Of the eighteen authors, five reported having financial relationships with vaccine manufacturers, and one reported a conflict of interest for serving as a consultant to the ACIP.[63] The lead author, William Thompson, has also publicly admitted that he and his coauthors “omitted statistically significant information” in an earlier study published in Pediatrics in 2004 that “suggested that African American males who received the MMR vaccine before age 36 months were at increased risk for autism.”[64]
In sum, other problems aside, the CDC cites this study as supporting its claims, but since it wasn’t designed to test the hypothesis that vaccines can cause autism, it doesn’t support the CDC’s claims that they do not. Its confirmation of an association between thimerosal exposure and tics also provides evidence against the CDC’s claim that thimerosal in vaccines is non-toxic.
The second relevant study from the list was written by A.W. McMahon et al. and published in the journal Vaccine in 2008. They specifically considered the use of thimerosal in influenza vaccines. But this was just another study looking at VAERS data that only looked at reports of “injection site reactions, rash, and infections” and didn’t even consider the question of autism. In fact, the word “autism” does not even appear in the study.[65]
In other words, it’s just another case of the CDC citing a study to support a claim that the study absolutely does not support. Since it was not designed to look for a possible association between the flu shot and autism, it cannot be cited as evidence of no association. It would be superfluous to examine this one further.
The third study was written by Cristofer S. Price and coauthors (including William Thompson) and published in Pediatrics in 2010. They compared 256 children exposed to thimerosal who had autism with 752 children exposed to thimerosal who did not have autism. So once again their study population was too small to be able to detect rare cases of autism caused by thimerosal-containing vaccines. They found that thimerosal exposures were similar between the two groups, but they did not compare autism rates between thimerosal-exposed children and children who received no exposure from vaccines. Once again, they did separate analyses for prenatal and postnatal exposures rather than considering total cumulative exposure. Once again, rather than considering the possibility of genetic or environmental susceptibility to vaccine injury manifesting as symptoms of autism, the authors excluded children with medical conditions known to be associated with an increased risk of autism. Once again, they acknowledged the inherent weakness of observational studies, including the risk of selection bias, and explicitly stated that no observational study “can definitively establish or disprove the hypothesis that thimerosal exposure increases the risk of ASDs [autism spectrum disorders].” To do that, they observed, would require “a large-scale prospective randomized trial”, which, they opined, was “not ethically feasible”.[66]
That admission, of course, does not stop the CDC from falsely citing this study as proof that vaccines do not cause autism.
The fourth study was written by John P. Barile and coauthors (including William Thompson) and published in the Journal of Pediatrics Psychology in 2011. Using a different statistical method, it reanalyzed data from the 2007 Thompson et al. study and confirmed the finding of a statistically significant association between thimerosal exposure and tics in boys. Since it looked at only seven outcomes compared to the original study’s forty-two outcomes, “it was not possible to rule out several of the other statistically significant associations from the previous study”. The authors also admitted that, “because this study excluded subjects born with a low birth weight and other confounding medical conditions, we may have excluded the children who were most vulnerable to the effects of thimerosal exposure.”[67] (Emphasis added.)
In sum, relevant to our purposes here, none of these studies demonstrate that vaccinating pregnant women with mercury-containing flu shots is safe for the developing fetus. Not one of them offers even the slightest shred of evidence that this practice of deliberately exposing fetuses to a known neurotoxin is not contributing to the alarming increasing rate of autism in the US, which was estimated in a 2018 study to now be one in forty children. Naturally, studies that aren’t designed to test a hypothesis cannot possibly disprove it.[68]
“Vaccines Do Not Cause Autism”?
As we’ve just seen, to support its assertion that science has proven that don’t cause autism, the CDC cites observational studies that, by CDC researchers’ own admission, can neither prove nor disprove a causal relationship, while also refusing to do the type of study that could.
Moreover, on March 29, 2008, CDC Director Julie Gerberding admitted on CNN, “Now, we all know that vaccines can occasionally cause fevers in kids. So if a child was immunized, got a fever, had other complications from the vaccines, and if you’re predisposed with a mitochondrial disorder, it can certainly set off some damage. Some of the symptoms can be symptoms that have characteristics of autism.”[69] (Emphasis added.)
She went on to emphasize that studies had found no association between vaccination and autism, but it’s important to understand that Gerberding’s phrase “characteristics of autism” is synonymous with “autism”, inasmuch as autism is a disease diagnosed on the basis of its symptoms. Furthermore, Gerberding was referring specifically to the case of Hannah Poling, a girl who had developmentally regressed into diagnosed autism after receiving nine vaccine doses at once at 19 months of age.
The Poling family is among those who have been awarded compensation by the government under the Vaccine Injury Compensation Program. There have been many other compensated cases in which the injured child had autism. According to a 2011 study published in Pace Environmental Law Review, “the VICP has been compensating cases of vaccine-induced encephalopathy and residual seizure disorder associated with autism since the inception of the program.” The authors found “eighty-three cases of autism among those compensated for vaccine-induced brain damage.”[70]
With respect to young Hannah, the government conceded that “the facts of this case meet the statutory criteria for demonstrating that the vaccinations CHILD received on July 19, 2000, significantly aggravated an underlying mitochondrial disorder, which predisposed her to deficits in cellular energy metabolism, and manifested as a regressive encephalopathy with features of autism spectrum disorder.”[71] (Emphasis added.)
One of the CDC’s top researchers is Dr. Frank DeStefano, the longtime director of the agency’s Immunization Safety Office and a coauthor of three of the nine aforementioned studies that the CDC cites to support its claim that vaccines do not cause autism. He was also a coauthor on five of the seven studies cited by the CDC to support its claim that it’s safe for pregnant women to get a flu shot. In a 2014 interview with journalist Sharyl Attkisson, DeStefano acknowledged that “it’s a possibility” that vaccines could trigger autism in genetically susceptible individuals. The problem, he elaborated, is that it is very “hard to predict who those children might be”, and trying to determine what underlying conditions put children at greater risk of being injured by vaccines is “very difficult to do”.[72] (Emphasis added.)
One of the reviewers of the 2004 IOM report that the CDC cites to support its claim that vaccines don’t cause autism was Dr. Andrew Zimmerman, a pediatric neurologist, associate professor of neurology and psychiatry at the Johns Hopkins University School of Medicine, and director of medical research at the Kennedy Krieger Institute’s Center for Autism and Related Disorders. Zimmerman has also been an expert witness for the government in cases under the Vaccine Injury Compensation Program.
In 2007, Zimmerman served as an expert witness for a case in which the petitioners were claiming that vaccination caused autism in a girl named Michelle Cedillo. In written testimony, Zimmerman expressed his professional opinion that there was no scientific basis for concluding that it was vaccination that caused her autism. He subsequently spoke with the government’s attorneys to clarify that this opinion applied only to that particular case and could not be generalized to other cases. In a sworn affidavit notarized on September 7, 2018, Zimmerman has testified (emphasis added):
I explained that I was of the opinion that there were exceptions in which vaccinations could cause autism.
More specifically, I explained that in a subset of children with an underlying mitochondrial dysfunction, vaccine induced fever and immune stimulation that exceeded metabolic energy reserves could, and in at least one of my patients, did cause regressive encephalopathy with features of autism spectrum disorder.
I explained that my opinion regarding exceptions in which vaccines could cause autism was based upon advances in science, medicine, and clinical research of one of my patients in particular.
The exceptional patient he was referring to was Hannah Poling.
Shortly after clarifying his opinion to the Department of Justice attorneys, he was contacted and informed that he “would no longer be needed as an expert witness”.
But that’s not all.
Despite having been informed directly by Dr. Zimmerman that his opinion in the Cedillo case could not be generalized to other cases, and that in some cases vaccines can cause autism, the lead attorney for the Department of Health and Human Services, Vincent Matanoski, cited Zimmerman’s opinion in the Cedillo case to support the government’s argument that vaccines did not cause autism in the case of a child named William “Yates” Hazlehurst. After reading Zimmerman’s testimony for the Cedillo case, Matanoski drew his point to a close by saying, “We know his views on this issue.”
In other words, the government’s lead attorney knowingly and willfully lied to deny that vaccination can cause autism.
Dr. Zimmerman’s true opinion on the Hazlehurst case, arrived at after reviewing the boy’s medical records, is that he “suffered regressive encephalopathy with features of autism spectrum disorder as a result of a vaccine injury”.
As Dr. Zimmerman continues in his affidavit (emphasis added):
In my opinion, the statement by Mr. Matonoski [sic] during his closing argument regarding my expert opinion was highly misleading and not an accurate reflection of my opinion for two reasons. First, Mr. Matanoski took portions of my opinion out of context. My opinion as to Michelle Cedillo was case specific. I was only referring to the medical evidence that I had reviewed regarding her. My opinion regarding Michelle Cedillo was not intended to be a blanket statement as to all children and all medical science. Second, as explained above, I specifically explained to Mr. Matanoski and the other DOJ attorneys who were present that there were exceptions in which vaccinations could cause autism.[73]
In his written testimony for the Cedillo case, Dr. Zimmerman also noted that one focus of autism research is the potential for maternal immune activation during pregnancy to cause autism. “Indeed, a current focus of immune research is on the mother’s immune system, which shows that maternal antibodies directed to the developing human fetal brain may be pathogenic (cause pathology) before birth.”[74]
As a 2003 paper written by an FDA researcher and published in the journal Vaccine noted, concerns about vaccinating pregnant women include “potential adverse effects caused by the immune response induced by the vaccine. While the relative exposure to the vaccine antigen may be very short, the induced maternal immune response is likely to persist and there is concern that it may affect the development of the fetus as well as its developing immune system leading to potential congenital and/or functional abnormalities. Moreover, immune modulation in the mother caused by vaccination during pregnancy could theoretically have an adverse influence on pregnancy outcome.” (Emphasis added.)
Coupled with this is the concern that “there is no data to address developmental risk in pregnant women or women of reproductive age at the time of licensure.”[75]
A study published in Vaccine in 2011 also highlighted a lack of data on the maternal inflammatory response to vaccination and acknowledged the concern that greater inflammatory responses may increase the risk of adverse outcomes. While influenza infection during pregnancy presents this risk to the developing fetus, “the inflammatory response to vaccination may itself be detrimental to fetal brain development.” (Emphasis added.)
The authors pointed out there is “substantial immune adaptation that occurs during pregnancy, with reported down-regulation of inflammatory responding”. In other words, there is naturally an anti-inflammatory environment for the fetus. But their study showed “that seasonal influenza virus vaccination elicits a significant inflammatory response among pregnant women.” While describing the vaccine-caused inflammation as “considerably milder and more transient than that elicited by infection”, there was also “substantial variability in the magnitude of response to vaccination.”[76]
In other words, certain women who have a more inflammatory response to vaccination may have a higher risk of adverse outcomes for their child from getting a flu shot during pregnancy.
Furthermore, it is not a simple trade-off between either getting a flu shot or getting the flu. While the authors compared the magnitude and duration of the inflammatory response from influenza infection with that from vaccination, they failed to consider that during any given flu season, most of the population is not infected with influenza.
According to a CDC study published in Vaccine, only about 8 percent of the US population on average is infected with influenza annually.[77]
A 2017 study published in JAMA Pediatrics by Ousseny Zerbo et al. found that, in a cohort of 196,929 children born between 2000 and 2010, expectant mothers were diagnosed with influenza in only 1,400 cases—just 0.7 percent. (And most of those cases were not laboratory confirmed so could very well have been caused by other viruses.)[78]
It is not only the magnitude and duration of the inflammatory response from infection versus that from vaccination that needs to be considered. The authors of the 2011 Vaccine study were ignoring the fact that the CDC’s recommendation effectively advocates that pregnant women trade the relatively small potential risk of getting a fever from the flu with the virtually guaranteed risk of an inflammatory response to vaccination.
We should also keep in mind that, as we learned in part three, studies have shown that getting an annual flu shot can actually increase your risk of influenza and other viral infections.
A Found Association between the Flu Shot and Autism
The main purpose of study by Zerbo et al. in JAMA Pediatrics was to investigate whether there is an association between either influenza infection or vaccination during pregnancy and the risk of autism. Despite it being the only study, to my knowledge, that has examined the question of whether getting a flu shot during pregnancy can increase the child’s risk of autism, the CDC does not list it among the studies it cites to support its companion claims that flu shots are safe for pregnant women and vaccines don’t cause autism.
This shouldn’t be too surprising given the researchers’ findings. They found that, consistent with previous studies, influenza infection was not associated with an increased risk of autism. However, influenza vaccination during the first trimester “was associated with an increased risk”, indicating four additional cases of autism for every 1,000 women vaccinated.
The authors noted that, like infection, “influenza vaccination during pregnancy has been reported to induce a transient increase in the levels of a number of proinflammatory cytokines”. They also reported that the association between vaccination and autism lost statistical significance “after statistical correction for multiple comparisons.”[79]
Other researchers, however, have criticized Zerbo et al. for the method they used to “correct” the data, known as the Bonferroni correction. In a letter published in JAMA Pediatrics, Dr. Brian S. Hooker criticized the study for “inappropriate” use of this method since it assumes independence of the associations, whereas in the study “the associations were highly interdependent”.[80]
In statistics, a “type I error” is when a study finds an association that doesn’t exist (essentially, a false positive result). A “type II error” is when a study fails to reject a false null hypothesis (fails to find an association that does exist, or a false negative). The Bonferroni correction is intended to avoid the type I error, which in the case of Zerbo et al. means that they “corrected” the data ostensibly to avoid finding an unreal association. To do this, the authors identified numerous “confounding factors”, meaning other factors that could explain a spurious association between the flu shot and autism. They treated these variables as competing hypotheses and divided the risk among the total number of hypotheses, thereby reducing the found association between the flu shot and autism to statistical nonsignificance.
Variables treated by Zerbo et al. as “confounders” included year of conception; child’s sex; gestational age; maternal prepregnancy body mass index (BMI); maternal age at delivery; maternal education at delivery; maternal race or ethnicity; gestational diabetes; and maternal asthma, autoimmune disease, or allergies.
But as Dr. James Lyons-Weiler, Bernadette Pajer, and Zoey O’Toole explain in a detailed critique of the study published on the website Age of Autism, “A statistically significant but unwanted positive association is easy to hide by adding several additional proposed confounders, and treating them as independent ‘hypotheses’, when they are not, to justify invoking correction of the type 1 error risk for multiple comparisons. One can make a statistically significant finding vanish in a sea of statistical shamwizardy.”
The fundamental problem with the use of the Bonferroni correction by Zerbo et al. is that “the covariates used are not clearly independent of each other”. Instead, “Many of the variables treated as ‘confounders’ by Zerbo et al. may instead be useful predictor variables to understand specifically who may be at risk of serious adverse events after vaccination when used in combination. The variables are potential risk factors or risk indicators of environmental and/or genetic susceptibility that may interact with vaccination.”[81] (Bold emphasis added; italics in original.)
It’s possible, for example, that women with autoimmune disease are at a higher risk of influenza vaccination causing neurodevelopmental harm to their fetus, yet the “correction” used by Zerbo et al. presupposed that this can’t be true. And what their data showed without such unfounded presuppositions was that influenza vaccination during pregnancy is associated with an increased risk of autism.
In another response letter published in JAMA Pediatrics, a team of Italian researchers likewise criticized Zerbo et al. for having “used the Bonferroni correction, which only adjusts for independent comparisons”, while it was doubtful that some of their supposed confounders could be independent from each other.
They further noted that, according to the 2010 Cochrane review (discussed in part one of this series), for every 100 healthy adults vaccinated, only one to three cases of influenza are prevented, meaning that anywhere from 33 to 100 people need to be vaccinated in order for one to see a benefit. And data presented by Zerbo et al. indicated that only two per every 1,000 women in their study population experienced influenza infection, whereas 297 per every 1,000 were vaccinated. Hence vaccination likely produced more inflammatory effects than influenza among the study population.
They further criticized Zerbo et al. for concluding that, although further studies need to be done, no change in public vaccine policy is presently needed. “Instead,” the Italian researchers reasoned, “given the possible risk of 4 additional autism spectrum disorder cases for every 1000 women vaccinated, it seems reasonable to apply the precautionary principle and refrain from vaccinating women or at least to avoid vaccination in the first trimester of pregnancy.”[82]
It also seems reasonable to advise women that, if they are going to get a flu shot, they should get a thimerosal-free version—yet as we’ve seen, the CDC, having staked its reputation on the claim that thimerosal in vaccines is non-toxic, refuses to apply the precautionary principle even to this small extent.
One of the authors of that letter, Alberto Donzelli, has further discussed the JAMA Pediatrics study in a paper published in the International Journal of Environmental Research and Public Health. Donzelli writes that the adjustment method used by Zerbo et al., “only adjusts for independent comparisons that they arbitrarily elevated to eight.” While their study’s main analysis examined autism risk for vaccination at any time during pregnancy, “it is biologically plausible that eventual effects on the fetal nervous system occur in the first trimester, during embryogenesis, and therefore it would have been logical to perform the main analysis in the first trimester.”
Zerbo and his colleagues were notified of these criticisms and in a reply agreed “that eight independent comparisons were not justified, but again arbitrarily reduced them to six. They could legitimately have reduced the independent comparisons to three, one for each trimester of pregnancy: this logical correction would have maintained statistical significance of the excess ASD risk in the first trimester, even on adopting the Bonferroni adjustment which is more conservative than other equally legitimate methods of correction for multiple testing.” (Emphasis added.)
Citing a more recent 2018 Cochrane review, Donzelli further noted that the estimated number of pregnant women who need to be vaccinated in order for one to receive a benefit is fifty-five. “Therefore, at a population level, the inflammatory effects of 100 vaccinated women should be compared with those caused in about two women with influenza illness.”
In order to reasonably assess this risk, Donzelli observed, randomized controlled trials with long-term follow-up are required: “Only these trials can avoid the possible flawed picture of observational studies where many of the benefits are only hypothesized, and possible series harms are underestimated, both for the healthy-vaccinees bias and for the known tendency of passive pharmacovigilance to underreporting of adverse events, even serious ones, but with a fairly high background rate.”[83]
How Maternal Immune Activation Might Increase the Risk of Autism
As we’ve seen, the 2005 Environmental Health Perspectives study on thimerosal toxicity cited by the CDC to support its claim of safety actually found that ethylmercury accumulates in the brain, where it breaks down into inorganic mercury, which is associated with an activation of microglia in the brain, which is among the markers of a neuroinflammatory process seen in people with autism.[84]
As the 2011 Vaccine study acknowledged, vaccination during pregnancy may cause an inflammatory response that is also detrimental to fetal brain development.[85]
The 2017 JAMA Pediatrics study by Zerbo et al. further showed that getting a flu shot during pregnancy causes maternal inflammation and found an association with autism equivalent to four additional cases per 1,000 pregnant women vaccinated (before using an inappropriate statistical method to “correct” the data in favor of the null hypothesis).
Among the inflammatory cytokines Zerbo et al. found to be increased by influenza vaccination is interleukin-6 (IL-6). As they noted, “Studies on mice found an association between high interleukin-6 levels during pregnancy and abnormal behavior and brain structure.”[86]
Interleukin-6 is also among a number of markers of inflammation shown to be associated with autism in a 2005 study published in Annals of Neurology. That study demonstrated “an active neuroinflammatory process” in the brains of autistic patients, which was “characterized by activation of microglia and astroglia”. Their findings indicated that “innate neuroimmune reactions play a pathogenic role in an undefined proportion of autistic patients”.
“An issue that remains unclear”, its authors stated, “is how and when microglia and astroglia become activated in the brain of autistic patients.” They proposed that it might result from “unknown factors” that disturb prenatal or postnatal development of the central nervous system.[87]
Among the authors of that study was Dr. Andrew W. Zimmerman, the expert who served as one of the government’s witnesses in vaccine injury cases until he informed government lawyers of his professional opinion that vaccines can cause autism in some individuals.
A 2007 mouse study published in Journal of Neuroscience noted that maternal immune activation generally “is detrimental to the developing brain” and showed that IL-6 was a “critical” factor in the pathway by which it “alters fetal brain development”. Maternal immune activation also results in an “alteration in gene expression in the brains of the offspring”, and IL-6 is “central to the process” by which it “causes long-term behavioral alterations in the offspring.”[88]
As noted in a 2012 study published in Biochimica et Biophysica Acta (BBA) – Molecular Basis of Disease, “Elevated IL-6 in autistic brains has been a consistent finding.” Its authors set out to investigate “the mechanisms by which IL-6 may be involved in the pathogenesisis of autism”. An increasing number of studies had suggested that “dysregulated immune responses may be involved”, and IL-6 “has also been suggested as a key mechanistic pathway in maternal immune activation that may be associated with autism.” Studying mouse brains, their own findings supported “a critical role of IL-6 elevation in modulating autism-like behaviors through impairments on synapse formation, dendritic spine development, as well as on neuronal circuit balance.”[89]
A 2013 study published in Annals of Neurology concluded that even subtle activation of inflammatory cytokines due to maternal immune activation “is sufficient for producing specific behavioral and seizure impairments in the offspring” and that “IL-6 is necessary and sufficient for producing autism in the offspring”.[90]
A 2017 study published in Brain, Behavior, and Immunity noted that maternal immune activation during pregnancy is a risk factor for autism and showed that it “results in acute inflammatory responses in the fetal brain.” The “immune dysregulation” it causes “persists postnatally”. Interleukin-6 “is both necessary and sufficient” for mediating its effects on the development of autism-related behavioral abnormalities. As the authors stated, “transient induction of IL-6 in the fetal brain, during critical period of neurodevelopment, appear to set in motion events that lead to adverse neuropathological and behavioral outcomes in adulthood.” Their study demonstrated that maternal immune activation “through placental IL-6 signaling leads to adverse effects in the offspring”, including “long-term changes in brain development and behavior.”[91]
A 2018 review published in Pharmaceuticals noted that studies had established that autism is associated with immune system dysfunction and a strong inflammatory state, reflected in the central nervous system as brain inflammation. It described increased expression of pro-inflammatory cytokines including IL-6 as possibly “the principle mechanism in the pathophysiology” of autism.[92]
Hence, far from science having proven autism cannot be caused by vaccines, what the science is actually telling us is that there are known biological mechanisms by which vaccines, including the flu shot, might cause autism.
One mechanism is that vaccination of pregnant women causes maternal immune activation, which results in increased inflammatory cytokines and is a recognized risk factor for autism.
Another mechanism is that neurotoxic mercury from vaccination can pass through the placental and blood-brain barriers, causing microglial activation that is characteristic of the neuroinflammatory process seen in autistic patients.
Together, these mechanisms, coupled with the CDC’s flu shot recommendation for pregnant women, might help explain the disturbing increase in autism incidence in the US. But the CDC doesn’t want you to know about this science. The CDC pretends this science does not exist. Instead, the CDC lies that science has conclusively proven that vaccines cannot cause autism, just it lies that science has demonstrated that getting a flu shot during pregnancy is “very safe” for both the expectant mother and the unborn child.
How the Media Serve the State, Not the Public
For their part, the mainstream media, instead of doing journalism in service to the public, engage in public policy advocacy in service to the state.
We’ve already seen in part one of this series how the New York Times blatantly lies about what its own cited sources have to say about the safety and effectiveness of the flu shot. And in part three, we saw how Washington Post reporter Lena H. Sun brazenly lies that no vaccines are added to the CDC’s routine childhood schedule until it has been studied for safety when given with all the other vaccines on the schedule.
When the CDC study was published finding an increased risk of miscarriage associated with receipt of pH1N1-containing flu shots two years in a row, Lena Sun reported about it for the Post.
Researchers had “found a hint of a possible link”, Sun wrote, thus obfuscating the fact that the study had found a statistically significant association with vaccinated women having a 7.7 times greater odds of miscarriage. Sun withheld that odds ratio from Post readers.
She also falsely reported that this 2017 study was “the first to identify a potential link between miscarriage and the flu vaccine”, thus writing the earlier study by former CDC researcher Gary Goldman out of existence—relegating it down the memory hole, to borrow from George Orwell’s dystopian novel 1984.
She correctly reported that the study suggested “an association, not a causal link”, but then immediately quoted a CDC official describing it as finding “a possible link”, thus once again obfuscating the fact that the study did find a strong statistically significant association. (That is to say, it found a link, and possibly a causal one.)
Sun proceeded to relay the message from “experts” that getting a flu shot during pregnancy is “the most effective strategy to protect newborns” since the vaccine “is not approved for use in infants younger than six months.” Citing the CDC and referring to the studies we’ve dissected above, she parroted the deception that “Many previous studies have shown that flu vaccines can be given safely during pregnancy, including numerous studies that found no link between flu vaccination and miscarriage.”
The CDC study had methodological limitations, Sun stressed, including the small number of women who’d had miscarriages and received the flu shot two years in a row, as well as a hypothetical selection bias in which women seeking care for miscarriages were more likely to have been vaccinated.
Toward the end of her article, Sun acknowledged the possibility of a causal relationship, relaying the authors’ speculation that the observed association, if real, might be due to the composition of the vaccines given during those flu seasons and the immunological response to them. Further studies were required, she noted, in order to address this concern, and the CDC had a study underway with results expected in late 2018 or in 2019. (As discussed above, the CDC has yet to publish this study, which did not look at those same flu seasons.)
The overall message conveyed to Post readers was that, yes, a study had found “a hint of a possible link” between the flu shot and miscarriage, but that we should not be too concerned about this and that pregnant women should continue getting an annual flu shot because we can rest assured that our children’s health is best left entrusted to the wisdom and decision-making powers of the CDC.[93]
That article once again highlights the institutionalized cognitive dissonance related to the subject of vaccinations. Again, we are supposed to regard observational studies finding no association between vaccination and adverse events as determinative, and yet when such studies do find an association, we are reminded that they do not establish causality and are prone to selection biases.
Shedding some light on the nature of Lena Sun’s reporting on vaccines, journalist Rob Wipond in an article published in The BMJ observed how her article, along with articles by Mike Stobbe of the Associated Press and Helen Branswell of STAT, downplayed the CDC study’s findings. Not one of their reports informed the public of the statistically significant 7.7 odds ratio. Instead, they all characterized the study as though its findings presented no serious cause for concern.
As it turned out, these three journalists were specifically selected by the CDC to help get its public relations message out to the public. Ahead of the study’s publication, its authors and the CDC “were concerned about the possible media coverage and public reaction”, so “instead of issuing a general press release, the authors invited three selected journalists for exclusive early access”. The three selected journalists were Stobbe, Branswell, and Sun.
“These were people that others [on the team] had had good history with,” the study’s lead author James Donahue told The BMJ. “We wanted to make sure that they got a full story on this, and carried the right messages—the messages that we think are important.” The first message Donahue identified was that the 7.7 odds ratio wasn’t very informative. The second was that pregnant women should still “get protected with the vaccine.”
In contrast to the CDC message conveyed to the public through mainstream journalists like Lena Sun, Chris Del Mar, a “professor of public health at Australia’s Bond University and coordinating editor of a Cochrane group that has conducted several systematic reviews of influenza vaccines”, emphasized to highly ineffective the flu shot is. “It’s a terrible vaccine”, Del Mar told The BMJ. “Its efficacy is tiny.”
Del Mar also pointed out that while fever from influenza infection has been associated with an increased risk of miscarriage, fever is also a known side effect of the vaccine. Most people contract influenza—as opposed to influenza-like illnesses caused by other viruses—only once every ten years. Furthermore, there are other things, like simple hand washing, “that may be much more effective than vaccination,” Del Mar told The BMJ.
Nevertheless, Wipond observed, none of the news articles by the CDC’s hand-picked journalists “reminded pregnant women of the importance of hand washing or other barrier methods.”[94]
Lena Sun relayed the CDC’s public relations message that for pregnant women to get a flu shot is “the most effective strategy to protect newborns” as though that had been scientifically proven. On its website, the CDC proclaims that “An annual seasonal flu vaccine is the best way to help protect against flu.”[95]
Sun is not alone in mindlessly parroting the CDC’s claim. The mainstream media do so frequently. But the reality is that when the CDC communicates this belief to the public as though it was a scientifically proven fact, it is once again lying.
We’ve already seen how the CDC and the media greatly exaggerate the flu shot’s benefits and downplay, deny, or ignore its potential risks. Further exposing the big lie that science has shown it to be “the best way” to prevent the flu will be the purpose of the forthcoming fifth and final installment of this multi-part influenza vaccine exposé.
References
[1] Centers for Disease Control and Prevention, “Pregnant Women & Influenza (Flu)”, CDC.gov, last reviewed February 12, 2019, accessed May 7, 2019, https://www.cdc.gov/flu/highrisk/pregnant.htm.
[2] Aaron E. Carroll, “A Link Between Alcohol and Cancer? It’s Not Nearly as Scary as It Seems”, New York Times, November 10, 2017, https://www.nytimes.com/2017/11/10/upshot/health-alcohol-cancer-research.html.
[3] Ibid.
[4] Gary S Goldman, “Comparison of VAERS fetal-loss reports during three consecutive influenza seasons”, Human & Experimental Toxicology, May 2013, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3888271/
[5] James G. Donahue et al., “Association of spontaneous abortion with receipt of inactivated influenza vaccine containing H1N1pdm09 in 2010-11 and 2011-12”, Vaccine, September 25, 2017, https://doi.org/10.1016/j.vaccine.2017.06.069.
[6] CDC, “Pregnant Women & Influenza (Flu)”, op. cit.
[7] Leroy E. Burney, “Influenza Immunization”, Statement by the US Public Health Service, Public Health Reports, October 1960, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1929542/.
[8] Peter N. Doshi, “Influenza: A Study of Contemporary Medical Politics”, Massachusetts Institute of Technology, 2011, https://dspace.mit.edu/handle/1721.1/69811.
Centers for Disease Control and Prevention, “Prevention and Control of Influenza: Recommendations of the Advisory Committee on Immunization Practices (ACIP)”, MMWR, May 28, 2004, https://www.cdc.gov/mmwr/preview/mmwrhtml/rr5306a1.htm.
[9] Robert A. Bednarczyk, Dzifa Adjaye-Gbewonyo, and Saad B. Omer, “Safety of influenza immunization during pregnancy for the fetus and the neonate”, American Journal of Obstetrics & Gynecology, September 2012, https://doi.org/10.1016/j.ajog.2012.07.002.
[10] Ibid.
[11] Unfortunately, when such cases do arise, justice is never done. Instead of holding responsible corporate executives liable, the government typically enters into plea agreements with pharmaceutical companies resulting merely in fines being paid. The pharmaceutical companies basically get a slap on the wrist, the government gets some additional funding from being paid fines representing some mere fraction of the profits made through sales of the product, and the pharmaceutical companies go on about their business practically undeterred from engaging in criminal fraud again. See: Eugene McCarthy, “A Call to Prosecute Drug Company Fraud as Organized Crime”, Working Paper, August 6, 2018, accessed on the website of the University of Illinois on December 18, 2018, https://www.uis.edu/legalstudies/wp-content/uploads/sites/12/2018/08/Pharma-RICO-For-Webpage.pdf.
[12] I’m quoting here specifically from the insert for GlaxoSmithKline’s Fluarix product, but this warning is standard in this or similar wording in all inactived influenza vaccine package inserts. GlaxoSmithKline Biologicals, FLUARIX (Influenza Vaccine) Package Insert, FDA.gov, accessed December 5, 2018, https://www.fda.gov/downloads/BiologicsBloodVaccines/Vaccines/ApprovedProducts/UCM335392.pdf. For the parent page linking to this document, see: https://www.fda.gov/BiologicsBloodVaccines/Vaccines/ApprovedProducts/ucm112850.htm.
[13] CDC, “Pregnant Women & Influenza”, op. cit.
[14] Centers for Disease Control and Prevention, “Flu Vaccine Safety and Pregnancy”, CDC.gov, last reviewed September 19, 2017, accessed May 7, 2019, https://www.cdc.gov/flu/highrisk/qa_vacpregnant.htm.
[15] Centers for Disease Control and Prevention, “Flu Vaccination & Possible Safety Signal”, CDC.gov, last reviewed September 13, 2017, accessed May 7, 2019, https://www.cdc.gov/flu/professionals/vaccination/vaccination-possible-safety-signal.html.
[16] Melissa Jenco, “ACIP discusses flu vaccine data, JEV recommendations”, AAP News, February 28, 2019, https://www.aappublications.org/news/2019/02/28/acip022719.
[17] Sandee LaMotte, “Flu shot will not cause a pregnant woman to miscarry, study says”, CNN, February 28, 2019, https://www.cnn.com/2019/02/27/health/flu-vaccine-pregnancy-safety-miscarriage/index.html.
[18] National Institutes of Health, PubMed.gov, search for “Donahue Belongia” conducted on April 17, 2019, https://www.ncbi.nlm.nih.gov/pubmed/?term=Donahue+Belongia.
[19] CDC, “Flu Vaccine Safety and Pregnancy”, op. cit.
[20] CDC, “Flu Vaccination & Possible Safety Signal”, op. cit.
[21] Pedro L. Moro, et al., “Adverse events in pregnant women following administration of trivalent inactivated influenza vaccine and live attenuated influenza vaccine in the Vaccine Adverse Event Reporting System, 1990 – 2009”, American Journal of Obstetrics and Gynecology, February 2011, https://doi.org/10.1016/j.ajog.2010.08.050.
[22] Pedro L. Moro, et al., “Adverse events following administration to pregnant women of influenza A (H1N1) 2009 monovalent vaccine reported to the Vaccine Adverse Event Reporting System”, American Journal of Obstetrics and Gynecology, November 2011, https://doi.org/10.1016/j.ajog.2011.06.047.
[23] Pedro Moro et al., “Surveillance of Adverse Events After Seasonal Influenza Vaccination in Pregnant Women and Their Infants in the Vaccine Adverse Event Reporting System, July 2010–May 2016”, Drug Safety, February 2017, https://doi.org/10.1007/s40264-016-0482-1.
[24] Keith B. Hoffman et al., “Stimulated Reporting: The Impact of US Food and Drug Administration-Issued Alerts on the Adverse Event Reporting System (FAERS)”, Drug Safety, September 26, 2014, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4206770/.
[25] Moro et al., “Surveillance of Adverse Events”, op.cit.
[26] Stephanie A. Irving, et al., “Trivalent Inactivated Influenza Vaccine and Spontaneous Abortion”, Obstetrics & Gynecology, January 1, 2013, https://www.ncbi.nlm.nih.gov/pubmed/23262941.
[27] CDC, “Flu Vaccine Safety and Pregnancy”, op. cit.
[28] Ibid.
[29] Elyse Olshen Kharbanda et al., “Inactivated Influenza Vaccine During Pregnancy and Risks for Adverse Obstetric Events”, Obstetrics & Gynecology, September 2013, https://dx.doi.org//10.1097/AOG.0b013e3182a1118a.
[30] James D. Nordin et al., “Maternal Influenza Vaccine and Risks for Preterm or Small for Gestational Age Birth”, The Journal of Pediatrics, May 2014, https://doi.org/10.1016/j.jpeds.2014.01.037.
[31] Elyse Olshen Kharbanda, “First Trimester Influenza Vaccination and Risks for Major Structural Birth Defects in Offspring”, The Journal of Pediatrics, August 2017, https://doi.org/10.1016/j.jpeds.2017.04.039.
[32] CDC, “Flu Vaccine Safety and Pregnancy”, op. cit.
[33] Centers for Disease Control and Prevention, “Thimerosal in Flu Vaccine”, CDC.gov, last reviewed October 16, 2015, accessed May 7, 2019, https://www.cdc.gov/flu/prevent/thimerosal.htm.
[34] Leslie K. Ball, Robert Ball, and R. Douglas Pratt, “An Assessment of Thimerosal Use in Childhood Vaccines”, Pediatrics, May 5, 2001, https://doi.org/10.1542/peds.107.5.1147.
[35] CDC, “Thimerosal in Flu Vaccine”, op. cit.
[36] Centers for Disease Control and Prevention, “Thimerosal in Vaccines”, CDC.gov, updated October 27, 2015 and accessed December 18, 2018, https://www.cdc.gov/vaccinesafety/concerns/thimerosal/index.html.
[37] Ball, et al., op. cit.
[38] CDC, “Thimerosal in Vaccines”, op. cit.
[39] Ball, et al., op. cit.
[40] Ball, et al., op. cit.
[41] Andrea Rock, “Toxic Tipping Point”, Mother Jones, March/April 2004, https://www.motherjones.com/politics/2011/02/toxic-tipping-point/. Robert F. Kennedy, Jr., and Lyn Redwood, “CDC Knew Its Vaccine Program Was Exposing Children to Dangerous Mercury Levels Since 1999”, EcoWatch, January 18, 2017, https://www.ecowatch.com/cdc-mercury-vaccines-kennedy-2199157054.html. When accessed on December 21, 2018, this article’s URL was returning a 404 “not found” error, but a Google cached version was still accessible at: https://webcache.googleusercontent.com/search?q=cache:TqRm2FlU4hQJ:https://www.ecowatch.com/cdc-mercury-vaccines-kennedy-2199157054.html+&cd=1&hl=en&ct=clnk&gl=us. See also the linked file cited in the article titled “Review of Thimerosal by the Food and Drug Administration as Required by the Food and Drugs Adminstration Moderniztion Act (1997-199)”, accessed December 21, 2018; https://worldmercuryproject.org/wp-content/uploads/2016/11/Review_-Thimerosal_-FDARequired__FDA_Modernization_Act.pdf. The full text of the quote I have used here is cited from: US House of Representatives, Mercury in Medicine—Taking Unnecessary Risks, Congressional Record, Volume 149, Number 76, May 21, 2003, https://www.govinfo.gov/content/pkg/CREC-2003-05-21/html/CREC-2003-05-21-pt1-PgE1011-3.htm.
[42] Kennedy and Redwood, “CDC Knew”, op. cit. See also the linked Rumack models obtained through a FOIA request, “Ethyl mercury body burden after Thiomersal injections vs. Methyl mercury oral suggested limits”, World Mercury Project, accessed December 21, 2018, https://worldmercuryproject.org/wp-content/uploads/foia-rumack-mercury-models.pdf. Also: Robert F. Kennedy, Jr., “How the FDA Responds When Asked To Prove That IT’s Safe to Inject Mercury (Thimerosal) Into Babies”, Collective Evolution, November 16, 2017, https://www.collective-evolution.com/2017/11/16/how-the-fda-responds-when-asked-to-prove-that-its-safe-to-inject-mercury-thimerosal-into-babies/.
[43] David A. Geier and Mark R. Geier, “An assessment of the impact of thimerosal on childhood neurodevelopmental disorders”, Pediatric Rehabilitation, April–June 2003, https://www.tandfonline.com/doi/abs/10.1080/1363849031000139315.
[44] Kennedy and Redwood, “CDC Knew”, op. cit. Kennedy, “How the FDA Responds”, op. cit.
[45] Kennedy and Redwood, “CDC Knew”, op. cit. Kennedy, “How the FDA Responds”, op. cit. See also the linked copy of the email obtained through a FOIA request: From Leslie Ball to Norman Baylor, “RE: CDC Q and A’s”, July 6, 1999, https://worldmercuryproject.org/wp-content/uploads/foia-leslie-ball-fda-no-safe-level-of-mercury.pdf. Note that the email is marked as “Importance: High” and “Sensitivity: Confidential”.
[46] CDC, “Thimerosal in Vaccines”, op. cit. Centers for Disease Control and Prevention, “Understanding Thimerosal, Mercury, and Vaccine Safety”, CDC.gov, dated February 2013, accessed May 7, 2019, https://www.cdc.gov/vaccines/hcp/patient-ed/conversations/downloads/vacsafe-thimerosal-color-office.pdf. Institute of Medicine (IOM), Immunization Safety Review Committee, Immunization Safety Review: Vaccines and Autism (Washington, DC: National Academies Press, 2004), pp. 135, 136, 138; https://www.nap.edu/catalog/10997/immunization-safety-review-vaccines-and-autism. The IOM was reformed in 2015 as the National Academy of Medicine.
[47] Centers for Disease Control and Prevention, “Frequently Asked Questions about Thimerosal”, CDC.gov, updated August 28, 2015 and accessed December 20, 2018, https://www.cdc.gov/vaccinesafety/concerns/thimerosal/faqs.html. Centers for Disease Control and Prevention, “Thimerosal Publications and References”, CDC.gov, updated October 27, 2015, and accessed December 21, 2018, https://www.cdc.gov/vaccinesafety/concerns/thimerosal/publications.html.
[48] Ball, et al., op. cit.
[49] Robert J. Mitkus, et al., “A Comparative Pharmocokinetic Estimate of Mercury in U.S. Infants Following Yearly Exposures to Inactivated Influenza Vaccines Containing Thimerosal”, Risk Analysis, October 10, 2013, https://doi.org/10.1111/risa.12124.
[50] Thomas M. Burbacher et al., “Comparison of Blood and Brain Mercury Levels in Infant Monkeys Exposed to Methylmercury or Vaccines Containing Thimerosal”, Environmental Health Perspectives, April 21, 2005, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1280342/.
[51] G. Jean Harry, Martha W. Harris, Leo T. Burka, “Mercury concentrations in brain and kidney following ethylmercury, methylmercury and Thimerosal administration to neonatal mice”, Toxicology Letters, September 18, 2004, https://doi.org/10.1016/j.toxlet.2004.07.014.
[52] Gianpaolo Guzzi, et al., “Effect of thimerosal, methylmercury, and mercuric chloride in Jurkat T Cell Line”, Interdisciplinary Toxicology, September 2012, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3600517/.
[53] Antonio R. Gasset, Motokazu Itoi, Yasuo Ishii, “Teratogenicities of Ophthalmic Drugs: II. Teratogenicities and Tissue Accumulation of Thimerosal”, Archives of Ophtalmology, 1975, https://doi.org/10.1001/archopht.1975.01010020056010.
[54] José G. Dórea, “Integrating Experimental (In Vitro and In Vivo) Neurotoxicity Studies of Low-dose Thimerosal Relevant to Vaccines”, Neurochemical Research, June 2011, https://doi.org/10.1007/s11064-011-0427-0.
[55] C. M. Taylor, J. Golding, and A. M. Emond, “Lead, cadmium and mercury levels in pregnancy: the need for international consensus on levels of concern”, Journal of Developmental Origins of Health and Disease, February 2014, https://doi.org/10.1017/S2040174413000500.
[56] Keith Schofield, “The Metal Neurotoxins: An Important Role in Current Human Neural Epidemics?” International Journal of Environmental Research and Public Health, December 2017, https://dx.doi.org/10.3390/ijerph14121511.
[57] Centers for Disease Control and Prevention, “Vaccines Do Not Cause Autism”, CDC.gov, updated November 23, 2015 and accessed December 21, 2018, https://www.cdc.gov/vaccinesafety/concerns/autism.html.
[58] IOM, 2004, op. cit., pp. 4, 139.
[59] IOM, 2004, op. cit.
[60] Institute of Medicine, The Childhood Immunization Schedule and Safety (Washington, DC: National Academies Press, 2013), p. 6; https://www.nap.edu/catalog/13563/the-childhood-immunization-schedule-and-safety-stakeholder-concerns-scientific-evidence.
[61] CDC, “Vaccines Do Not Cause Autism”, op. cit.
[62] Centers for Disease Control and Prevention, “Science Summary: CDC Studies on Thimerosal in Vaccines”, accessed May 8, 2019, https://www.cdc.gov/vaccinesafety/pdf/cdcstudiesonvaccinesandautism.pdf.
[63] William W. Thompson et al., “Early Thimerosal Exposure and Neuropsychological Outcomes at 7 to 10 Years”, New England Journal of Medicine, September 27, 2007, https://doi.org/10.1056/NEJMoa071434.
[64] William W. Thompson, “Statement of William W. Thompson, Ph.D., Regarding the 2004 Article Examining the Possibility of a Relationship Between MMR Vaccine and Autism”, Morgan Verkamp LLC, August 27, 2014, https://www.morganverkamp.com/; archived April 1, 2016 at https://morganverkamp.com/statement-of-william-w-thompson-ph-d-regarding-the-2004-article-examining-the-possibility-of-a-relationship-between-mmr-vaccine-and-autism/.
[65] A.W. McMahon et al., “Inactivated influenza vaccine (IIV) in children <2 years of age: Examination of selected adverse events reported to the Vaccine Adverse Event Reporting System (VAERS) after thimerosal-free or thimerosal-containing vaccine”, Vaccine, January 17, 2008, https://doi.org/10.1016/j.vaccine.2007.10.071.
[66] Cristofer S. Price et al., “Prenatal and Infant Exposure to Thimerosal From Vaccines and Immunoglobulins and Risk of Autism”, Pediatrics, October 2010, https://doi.org/10.1542/peds.2010-0309.
[67] John P. Barile et al., “Thimerosal Exposure in Early Life and Neuropsychological Outcomes 7–10 Years Later”, Journal of Pediatric Psychology, January/February 2012, https://doi.org/10.1093/jpepsy/jsr048.
[68] Michael D. Kogan et al., “The Prevalence of Parent-Reported Autism Spectrum Disorder Among US Children”, Pediatrics, December 2018, https://doi.org/10.1542/peds.2017-4161.
[69] CNN, House Call With Dr. Sanjay Gupta, March 29, 2008, https://transcripts.cnn.com/TRANSCRIPTS/0803/29/hcsg.01.html.
[70] Mary Holland et al., “Unanswered Questions from the Vaccine Injury Compensation Program: A Review of Compensated Cases of Vaccine-Induced Brain Injury”, Pace Environmental Law Review, 2011, https://digitalcommons.pace.edu/pelr/vol28/iss2/6/.
[71] David Kirby, “The Vaccine-Autism Court Document Every American Should Read”, Huffington Post, February 26, 2008, https://www.huffpost.com/entry/the-vaccineautism-court-d_n_88558.
[72] Sharyl Attkisson, “CDC: ‘Possibility’ that vaccines rarely trigger autism”, December 10, 2018, SharylAttkisson.com, https://sharylattkisson.com/2018/12/10/cdc-possibility-that-vaccines-rarely-trigger-autism/.
[73] Dr. Andrew Walter Zimmerman, Affidavit, September 7, 2018. Published by Sharyl Attkisson, “Dr. Andrew Zimmerman’s full Affidavid on alleged link between vaccines and autism that U.S. govt. covered up”, SharylAttkisson.com, January 6, 2019, https://sharylattkisson.com/2019/01/06/dr-andrew-zimmermans-full-affidavit-on-alleged-link-between-vaccines-and-autism-that-u-s-govt-covered-up/. See also: Sharyl Attkisson, “The Vaccination Debate”, Full Measure, January 6, 2019, https://fullmeasure.news/news/cover-story/the-vaccination-debate. Sharyl Attkisson, “How a pro-vaccine doctor reopened debate about link to autism”, The Hill, January 13, 2019, https://thehill.com/opinion/healthcare/425061-how-a-pro-vaccine-doctor-reopened-debate-about-link-to-autism. Children’s Health Defense, “Robert F. Kennedy, Jr. Demands the Office of the Inspector General and Congress Investigate Department of Justice for Fraud and Obstruction of Justice”, PRNewswire, January 14, 2019, https://www.prnewswire.com/news-releases/robert-f-kennedy-jr-demands-the-office-of-the-inspector-general-and-congress-investigate-department-of-justice-for-fraud-and-obstruction-of-justice-300777802.html.
[74] Dr. Andrew W. Zimmerman, Letter to US Department of Justice, “Re: Cedillo case”, April 24, 2007, accessible at https://www.autism-watch.org/omnibus/expert_reports/FF_Zimmerman_Report_98-916.pdf.
[75] Marion F. Gruber, “Maternal immunization: US FDA regulatory considerations”, Vaccine, July 28, 2003, https://doi.org/10.1016/S0264-410X(03)00357-8.
[76] Lisa M. Christian et al., “Inflammatory Responses to Trivalent Influenza Virus Vaccine Among Pregnant Women”, Vaccine, September 22, 2011, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3204610/.
[77] According to a 2007 CDC study, about 25 million cases of influenza occur annually in the US, which given the population at the time of about 300 million means that about only about 8 percent of the population on average will get the flu during a given flu season. Noelle-Angelique M. Molinari, et al., “The annual impact of seasonal influenza in the US: Measuring disease burden and costs”, Vaccine, June 28, 2007, https://doi.org/10.1016/j.vaccine.2007.03.046.
[78] Ousseny Zerbo, et al., “Association Between Influenza Infection and Vaccination During Pregnancy and Risk of Autism Spectrum Disorder”, JAMA Pediatrics, January 2, 2017, https://doi.org/10.1001/jamapediatrics.2016.3609.
[79] Ibid.
[80] Brian S. Hooker, “Influenza Vaccination in the First Trimester of Pregnancy and Risk of Autism Spectrum Disorder”, JAMA Pediatrics, June 2017, https://doi.org/10.1001/jamapediatrics.2017.0734.
[81] James Lyons-Weiler, Bernadette Pajer, and Zoey O’Toole, “Seven Major Flaws in Zerbo et al. (2016): Association Between Influenza Infection and Vaccination During Pregnancy and Risk of Autism Spectrum Disorder”, December 20, 2016, https://www.ageofautism.com/2016/12/seven-major-flaws-in-zerbo-et-al-2016-association-between-influenza-infection-and-vaccination-during.html.
[82] Alberto Donzelli, Alessandro Schivalocchi, and Alessandro Battaggia, “Influenza Vaccination in the First Trimester of Pregnancy and Risk of Autism Spectrum Disorder”, JAMA Pediatrics, June 2017, https://doi.org/10.1001/jamapediatrics.2017.0753.
[83] Alberto Donzelli, “Influenza Vaccinations for All Pregnant Women? Better Evidence Is Needed”, International Journal of Environmental Research and Public Health, September 18, 2018, https://dx.doi.org/10.3390/ijerph15092034.
[84] Burbacher et al., op. cit.
[85] Christian et al., op. cit.
[86] Zerbo et al., op. cit.
[87] Diana L. Vargas et al., “Neurological Activation and Neuroinflammation in the Brain of Patients with Autism”, Annals of Neurology, January 2005, https://doi.org/10.1002/ana.20315.
[88] Stephen E. P. Smith et al., “Maternal Immune Activation Alters Fetal Brain Development through Interleukin-6”, Journal of Neuroscience, October 3, 2007, https://doi.org/10.1523/JNEUROSCI.2178-07.2007.
[89] Hongen Wei et al., “Brain IL-6 elevation causes neuronal circuitry imbalances and mediates autism-like behaviors”, Biochimica et Biophysica Acta (BBA) – Molecular Basis of Disease, June 2012, https://doi.org/10.1016/j.bbadis.2012.01.011.
[90] Eduardo Pineda et al., “Maternal immune activation promotes hippocampal kindling epileptogenesis in mice”, Annals of Neurology, March 21, 2013, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3775928/.
[91] Wei-Li Wu et al., “The placental interleukin-6 signaling controls fetal brain development and behavior”, Brain, Behavior, and Immunity, May 2017, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5373986/.
[92] Dario Siniscalco et al., “Inflammation and Neuro-Immune Dysregulations in Autism Spectrum Disorders”, Pharmaceuticals, June 4, 2018, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6027314/.
[93] Lena H. Sun, “What to know about a study of flu vaccine and miscarriage”, Washington Post, September 13, 2017, https://www.washingtonpost.com/news/to-your-health/wp/2017/09/13/researchers-find-hint-of-a-link-between-flu-vaccine-and-miscarriage/.
[94] Rob Wipond, “Reporting flu vaccine science”, BMJ, January 5, 2018, https://doi.org/10.1136/bmj.k15.
[95] Centers for Disease Control and Prevention, “Key Facts About Seasonal Flu Vaccine”, CDC.gov, last reviewed September 6, 2018, accessed May 9, 2019, https://www.cdc.gov/flu/prevent/keyfacts.htm.




The flu shot at 4 months pregnant is the major reason I have been citing for my son’s autism. There were no less than three safety warnings on the insert from the manufacturer to DO NOT GIVE TO PREGNANT WOMEN. THREE!!! But that info is rendered superfluous because no one reads those things except for ME! Lord knows the doctors and nurses giving the vaccines NEVER read them! That is a confirmed fact from the birth of our last son, 19 months ago, Feb.,2019. Not one medical “professional was familiar with any of the safety inserts.
It also brought up another EXTREMELY salient point by mentioning under the adverse reactions that there IS A LINK TO AUTISM. So the manufacturers have already admitted in print that there is a link between vaccines and autism yet the public “debate” rages on because…………………….???
We are under attack by a triumvirate of media, government and Big Pharma. It is time to wake up and take off the gloves, for the sake of our children and society.
Yesterday I went to the CDC site to see what recommendations they have for Pregnant woman and they showed nearly all vaccines should be given to a pregnant woman…..so I called their 800 number and told them my relative, was given the Flu vax , and a ultrasound , at 3 months pg for her second baby. and she went in for a 4 month check up and her doctor told her the baby had no heart beat and had died and her OB doctor gave her a prescription for some pill to expell it and she nearly bled to death in the shower and had to the ER. her husband said.
Michigan LIve also published another report of a woman in Ionia ,Mi who had went through the same thing my relative did.
So,I told the CDC, that I worked in Labor and Delivery for years and mothers were told not to smoke or drink or take any pills during pregnancy and now they are giving a PG woman all of these vaccines ?
So by the time I got off the phone with them that CDC site was being froze and my internet froze up and I had to get off the internet and I tried to get back into the CDC site to that page I saw and it has disappeared ….
.I was irate and I told the CDC person who answered the phone all I could about the one million Pregnant Kenya woman miscarriaging after vaccinations at 3 months pg. …And that the CDC is only an advisory
So today , I woke up thinking I had to investigate Aluminum in these vaccines some more and sure enough for an hour and half I finally found one that has been missed by Del Bigtree,RFK jr, and all the rest of them that must not have researched it enough …
So here is another one of my contributions for FREE !
Read and post it because this study and many more studies I saw on tests with aluminum on sheep and what it caused an animal but this one is very conclusive Aluminum causes Brain Inflammation and Autism.
Conclusion
The elevated brain cytokines indicate that aluminum adjuvant can cause brain injuries by the same mechanisms as immune activation.
Aluminum adjuvant causes long term, chronic immune activation/inflammation in the brain, and such inflammation is a proven cause of neurodevelopmental disorders, including autism.
Aluminum Adjuvant and Brain Inflammation
Several studies show that injection of Al adjuvant causes inflammation and microglial activation in the central nervous system. None of these studies measured IL-6, unfortunately. But microglial activation suggests that IL-6 is elevated, since microglia can produce IL-6, and often do produce IL-6 when activated and/or exposed to oxidative stress.
Shaw et al, 2009: Aluminum hydroxide injections lead to motor deficits and motor neuron degeneration
https://vaccinepapers.org/aluminum-inflammation-interleukin-6/
I went trought same thing. I was pregnat 10 weeks. And obs told me to het the flue shot . Because was good for the future baby. Well i got the shot. And days later. Start bleeding, and went to emergency. And they told me Just the heart baby stop beeping. They said it was a simple miscarrige . No I blame the flue shot
Great work, as usual. You are a treasure.
Best,
Stacia
Thank you!